The Nathan K. Archer Laboratory
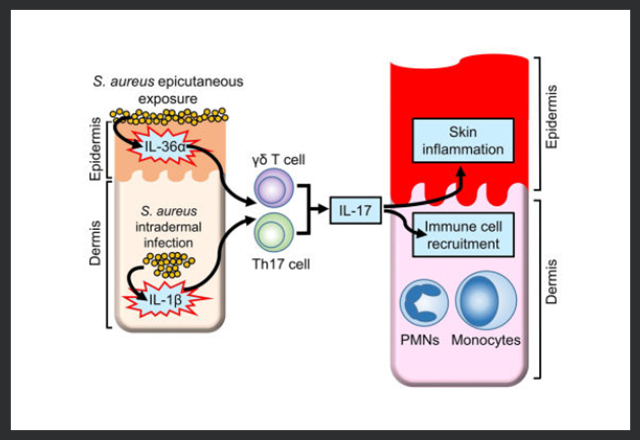 Staphylococcus aureus colonization during atopic dermatitis contributes to skin inflammation, but the underlying mechanisms are unclear. Our research demonstrates that epicutaneous S. aureus exposure drives skin inflammation, which is mediated by the bacterial PSMα and host IL-36R/MyD88-induced production of IL-17 by T cells.
Staphylococcus aureus colonization during atopic dermatitis contributes to skin inflammation, but the underlying mechanisms are unclear. Our research demonstrates that epicutaneous S. aureus exposure drives skin inflammation, which is mediated by the bacterial PSMα and host IL-36R/MyD88-induced production of IL-17 by T cells.Welcome to the Nathan K. Archer Laboratory at the Johns Hopkins Department of Dermatology.
Our goal is to understand mechanisms of protective innate and adaptive immune responses to skin pathogens and the role of aberrant immune responses and the skin microbiome in the pathogenesis of inflammatory skin diseases, including atopic dermatitis and psoriasis. We have made discoveries involving Toll-like receptors (TLRs), IL-1 family cytokines, inflammasome responses, and the role of different T cell subsets (especially IL-17-producing T cells). We are currently investigating the protective immunity to Staphylococcus aureus, which is the most common human bacterial skin pathogen. This area of research is highly significant since S. aureus infections represent a major public health threat due to the widespread emergence of virulent community-acquired methicillin-resistant S. aureus (MRSA) strains. In our work involving pathogenic immune mechanisms and dysbiosis of the skin microbiome in contributing to skin inflammation and allergic disease, we have made key discoveries involving the role of IL-36, MyD88-signaling, and STAT3-signaling. Our long-term goal is to discover mechanisms that can serve as targets for future immune-based therapies and vaccination strategies.
Our research has used innovative in vivo preclinical models, including pioneering advanced techniques of in vivo whole animal optical (bioluminescence and fluorescence) imaging to track bacterial clearance and host immune responses noninvasively and longitudinally over time, human skin organotypic culture models and novel humanized mouse models possessing both human skin and immune cells. Our laboratory has been funded with NIH grant support (R01 and K01 grants) and extensive industry and foundation support.
In addition to our work in the skin, we are investigating protective immune responses and novel antibacterial coatings to help treat or prevent bacterial biofilm-related infections of medical devices, prostheses, and implants. These infections are a devastating complication as bacterial biofilms form on the foreign implanted materials, which inhibit the efficacy of antibiotics and blocks penetration of immune cells. We have multiple preclinical animal models of these post-surgical infections using the latest technology in multimodality imaging, including optical, PET, CT, and photoacoustic imaging. The goal is to provide new insights into protective immune responses and novel treatments against these biofilm-related implant infections.
Selected Publications
Please review these selected publications or Dr. Archer's full bibliography.
-
- Alphonse MP, Rubens JH, Ortines RV, Orlando NA, Patel AM, Dikeman D, Wang Y, Vuong I, Joyce DP, Zhang J, Mumtaz M, Liu H, Liu Q, Youn C, Patrick GJ, Ravipati A, Miller RJ, Archer NK*, Miller LS*. 2021. Pan-caspase inhibition as a potential host-directed immunotherapy against MRSA and other bacterial skin infections. Science Translational Medicine. PMID: 34233954.
- Patrick GJ*, Liu H*, Alphonse MP, Dikeman DA, Youn C, Otterson JC, Wang Y, Ravipati A, Mazhar M, Denny G, Ortines RV, Zhang E, Miller RJ, Dillen CA, Liu Q, Nolan SJ, Nguyen K, Marcello L, Do DC, Wier EM, Zhang Y, Caviness G, Klimowicz AC, Mierz DV, Fine JS, Sun G, Goldbach-Mansky R, Marusina AI, Merleev AA, Maverakis E, Garza LA, Milner JD, Gao P, Ramanujam M, Raymond EL, Archer NK*, Miller LS*. 2021. Epicutaneous Staphylococcus aureus induces IL-36 to enhance IgE production and ensuing allergic disease. The Journal of Clinical Investigation. 131(5):143334. PMID: 33645549.
- Archer NK, Jo JH, Lee SK, Kim D, Smith B, Ortines RV, Wang Y, Marchitto MC, Ravipati A, Cai SS, Dillen CA, Liu H, Miller RJ, Ashbaugh AG, Uppal AS, Oyoshi MK, Malhotra N, Hoff S, Garza LA, Kong HH, Segre JA, Geha RS, Miller LS. 2018. Injury, dysbiosis and filaggrin deficiency drive skin inflammation via keratinocyte IL-1α release. Journal of Allergy & Clinical Immunology S0091-6749 (18): 31295-8. PMCID: PMC6424655.
- Advaitaa Ravipati, Sabrina Nolan, Martin Alphonse, Dustin Dikeman, Christine Youn, Yu Wang, Nicholas Orlando, Garrett Patrick, Steven Lee, Roger V. Ortines, Haiyun Liu, Robert J. Miller, Carly A. Dillen, Mark Marchitto, S. Sarah Cai, Lloyd S. Miller, Nathan K. Archer IL-6R/STAT3-signaling in keratinocytes rather than T cells induces psoriasis-like dermatitis in mice Journal of Investigative Dermatology
- Dong X, Limjunyawong N, Sypek EI, Wang G, Ortines RV, Youn C, Alphonse MP, Dikeman D, Wang Y, Lay M, Kothari R, Vasavda C, Pundir P, Goff L, Miller LS, Lu W, Garza LA, Kim BS, Archer NK*, Dong X*. Keratinocyte-derived defensins activate neutrophil-specific receptors Mrgpra2a/b to prevent skin dysbiosis and bacterial infection. Immunity. 2022 Sep 13;55(9):1645-1662.e7. doi: 10.1016/j.immuni.2022.06.021. Epub 2022 Jul 25. PubMed PMID: 35882236; PubMed Central PMCID: PMC9474599.
- Gordon O, Dikeman DA, Ortines RV, Wang Y, Youn C, Mumtaz M, Orlando N, Zhang J, Patel AM, Gough E, Kaushik A, Nuermberger EL, Upton AM, Fotouhi N, Miller LS, Archer NK. The Novel Oxazolidinone TBI-223 Is Effective in Three Preclinical Mouse Models of Methicillin-Resistant Staphylococcus aureus Infection. Microbiol Spectr. 2022 Oct 26;10(5):e0245121. doi: 10.1128/spectrum.02451-21. Epub 2022 Sep 15. PubMed PMID: 36106881; PubMed Central PMCID: PMC9603142.
- Wang Y, Dikeman D, Zhang J, Ackerman N, Kim S, Alphonse MP, Ortines RV, Liu H, Joyce DP, Dillen CA, Thompson JM, Thomas AA, Plaut RD, Miller LS, Archer NK. CCR2 contributes to host defense against Staphylococcus aureus orthopedic implant-associated infections in mice. J Orthop Res. 2022 Feb;40(2):409-419. doi: 10.1002/jor.25027.
-
- Patrick GJ, Archer NK, Miller LS. 2021. Which way do we go? Complex interactions in atopic dermatitis pathogenesis. Journal of Investigative Dermatology 141(2):274-284. PMCID: PMC7855294.
- Youn C, Archer NK, Miller LS. 2020. Research Techniques Made Simple: Mouse bacterial skin infection models for immunity research. Journal of Investigative Dermatology 140(8):1488-1497.e1. PMCID: PMC7387158
- Archer NK, Wang Y, Ortines RV, Liu H, Nolan SJ, Liu Q, Alphonse MP, Dikeman DA, Mazhar M, Miller RJ, Anderson LS, Francis KP, Simon SI, Miller LS. 2019. Preclinical models and methodologies for monitoring Staphylococcus aureus infections using noninvasive optical imaging. Methods in Molecular Biology 2069:197-228. PMCID: PMC7745539.
- Archer NK, Dilolli MN, Miller LS. 2017. Pushing the envelope in psoriasis: Late cornified envelope proteins possess antimicrobial activity. Journal of Investigative Dermatology 137 (11): 2257-2259. PMCID: PMC7727264.
Archer Lab Members
-
 Dr. Nathan K. Archer, Ph.D., earned his B.S. in Biochemistry at the University of Wisconsin, Madison and he earned his Ph.D. degree at the University of Maryland, Baltimore. Dr. Archer performed his post-doctoral research fellowship in the Johns Hopkins Department of Dermatology. Dr. Archer’s research involves the study of innate and adaptive immune responses in the skin. He has been an NIH-funded investigator since 2018. In 2018, Dr. Archer joined the Johns Hopkins Department of Dermatology as an Assistant Professor to continue his research involving cutaneous immunology, host-microbe interactions, and biofilm-related infections.
Dr. Nathan K. Archer, Ph.D., earned his B.S. in Biochemistry at the University of Wisconsin, Madison and he earned his Ph.D. degree at the University of Maryland, Baltimore. Dr. Archer performed his post-doctoral research fellowship in the Johns Hopkins Department of Dermatology. Dr. Archer’s research involves the study of innate and adaptive immune responses in the skin. He has been an NIH-funded investigator since 2018. In 2018, Dr. Archer joined the Johns Hopkins Department of Dermatology as an Assistant Professor to continue his research involving cutaneous immunology, host-microbe interactions, and biofilm-related infections.Over the years, Dr. Archer has given lectures about his research at the Society of Investigative Dermatology (SID) Annual Meetings in 2017 and 2019, the Immunology Annual Meeting in 2017, and at international scientific meetings, including the International Symposium on Staphylococci and Staphylococcal Infections (France, 2012) and the International Conference on Biofilms (China, 2019).
-
 Yu Wang received her Ph.D. at Institute of Hydrobiology, Chinese Academy of Sciences, P.R.China in 2006. Her graduate school work involved the gene regulation during cell differentiation and cell division with specific emphasis on the regulation of gene expression during heterocyst differentiation and cell division in Anabaena. From 2007 to 2013, she has received her postdoctoral training in Department of Cell Biology, Johns Hopkins University School of Medicine. During this period, her research interest had been focusing on the cellular function of protein Huntingtin and Rho family small GTPases in cell directed migration and cell cytokinesis. Yu joined the Archer lab in March 2013, and she has been working on using antimicrobial polymer and nanoparticle coatings to prevent orthopaedic implant infections, and investigating wound healing using in vivo models of wounding.
Yu Wang received her Ph.D. at Institute of Hydrobiology, Chinese Academy of Sciences, P.R.China in 2006. Her graduate school work involved the gene regulation during cell differentiation and cell division with specific emphasis on the regulation of gene expression during heterocyst differentiation and cell division in Anabaena. From 2007 to 2013, she has received her postdoctoral training in Department of Cell Biology, Johns Hopkins University School of Medicine. During this period, her research interest had been focusing on the cellular function of protein Huntingtin and Rho family small GTPases in cell directed migration and cell cytokinesis. Yu joined the Archer lab in March 2013, and she has been working on using antimicrobial polymer and nanoparticle coatings to prevent orthopaedic implant infections, and investigating wound healing using in vivo models of wounding.
-
 Sabrina received her B.Sc. in Biological Sciences at Edinburgh University, Edinburgh, U.K., and M.Sc. in Biology and Immunology at American University, Washington D.C. Her graduate work focused on the use of lipids by the intracellular parasite Toxoplasma gondii under Dr. Isabel Coppens at the Johns Hopkins Bloomberg School of Public Health. There, she earned her PhD in Molecular Microbiology and Immunology in 2016. Sabrina joined the Archer lab in September 2020 and will be working on the immune responses that promote atopic dermatitis skin inflammation and allergic responses upon S. aureus epicutaneous exposure.
Sabrina received her B.Sc. in Biological Sciences at Edinburgh University, Edinburgh, U.K., and M.Sc. in Biology and Immunology at American University, Washington D.C. Her graduate work focused on the use of lipids by the intracellular parasite Toxoplasma gondii under Dr. Isabel Coppens at the Johns Hopkins Bloomberg School of Public Health. There, she earned her PhD in Molecular Microbiology and Immunology in 2016. Sabrina joined the Archer lab in September 2020 and will be working on the immune responses that promote atopic dermatitis skin inflammation and allergic responses upon S. aureus epicutaneous exposure.
-
 Christine received her B.Sc. in Biology at Emory University College of Pharmacy, Atlanta and M.Sc. in Pharmacology at Seoul National University College of Pharmacy, Seoul, S. Korea. Her masters work focused on antitoxin and vaccine efficacy for three different types of biological toxins. Christine joined the Cellular and Molecular Medicine PhD program in 2018 and the Archer lab in 2019 for her graduate thesis work on understanding the host defense role of TNF and STAT3 against S. aureus skin infections.
Christine received her B.Sc. in Biology at Emory University College of Pharmacy, Atlanta and M.Sc. in Pharmacology at Seoul National University College of Pharmacy, Seoul, S. Korea. Her masters work focused on antitoxin and vaccine efficacy for three different types of biological toxins. Christine joined the Cellular and Molecular Medicine PhD program in 2018 and the Archer lab in 2019 for her graduate thesis work on understanding the host defense role of TNF and STAT3 against S. aureus skin infections.
-
- Garrett Patrick, B.S., Former Graduate Student 2018-2020. Current Position: Medical student, Johns Hopkins School of Medicine, Baltimore, MD.
- Advaitaa Ravipati, M.S., Former Research Technician 2018-2019. Current Position: Medical student, University of Miami, Miami, FL.
- Martin Prince Alphonse, M.Sc., Ph.D., Former Post-doctoral Fellow
- Dustin Dikeman, M.A., Former Research Specialist II
- Nicholas Orlando, B.S., Former Masters Student
Contact Us
Nathan K. Archer, Ph.D.
Assistant Professor of Dermatology
Faculty Member, Cellular and Molecular Medicine (CMM) and Immunology Graduate Programs
Phone (office): 410-614-3490
Email: [email protected]
Johns Hopkins University School of Medicine
Johns Hopkins Department of Dermatology
Office – Suite 2M04, Koch Cancer Research Building II
Lab – Suite 216, Koch Cancer Research Building II
1550 Orleans Street
Baltimore, MD 21231
Phone (lab): 410-614-4520
Fax: 410-955-8635
Twitter: @ArcherLab
