Translational Research and Innovation Laboratory (TRAIL)
Translational Research and Innovation Laboratory (TRAIL) was created in 2023 with the mission to bridge the gap between patients and laboratory science through teamwork and innovation.
The TRAIL is focused on answering questions raised by our clinical and clinical research colleagues. The capabilities of our laboratory are solely guided by the clinical and clinical and translational research needs of our Johns Hopkins All Children’s Hospital clinical investigators, as well as by investigators from collaborating institutions, with the aim to improve outcomes for all children.
The TRAIL team consists of laboratory-trained scientists and clinical investigators, and our success is based on leveraging a breadth of expertise and experience of individual team members, for robust solutions and innovations that would otherwise not be possible.
TRAIL leadership team
Vera Ignjatovic PhD, MBA
Professor (PAR) of Pediatrics

Neil Goldenberg MD, PhD
Professor of Pediatrics

Steven Bruzek MS, MB
Program Manager

TRAIL investigators
Marisol Betensky, MD, MPH
Assistant Professor of Pediatrics

John Morrison, MD, PhD
Assistant Professor of Pediatrics

Anthony Alexander Sochet, M.D., M.H.S.
Assistant Professor of Anesthesiology and Critical Care Medicine

Amy Kiskaddon PharmD, MBA
Assistant Professor of Pediatrics

Maha Al-Ghafry, M.D.
Assistant Professor of Pediatrics

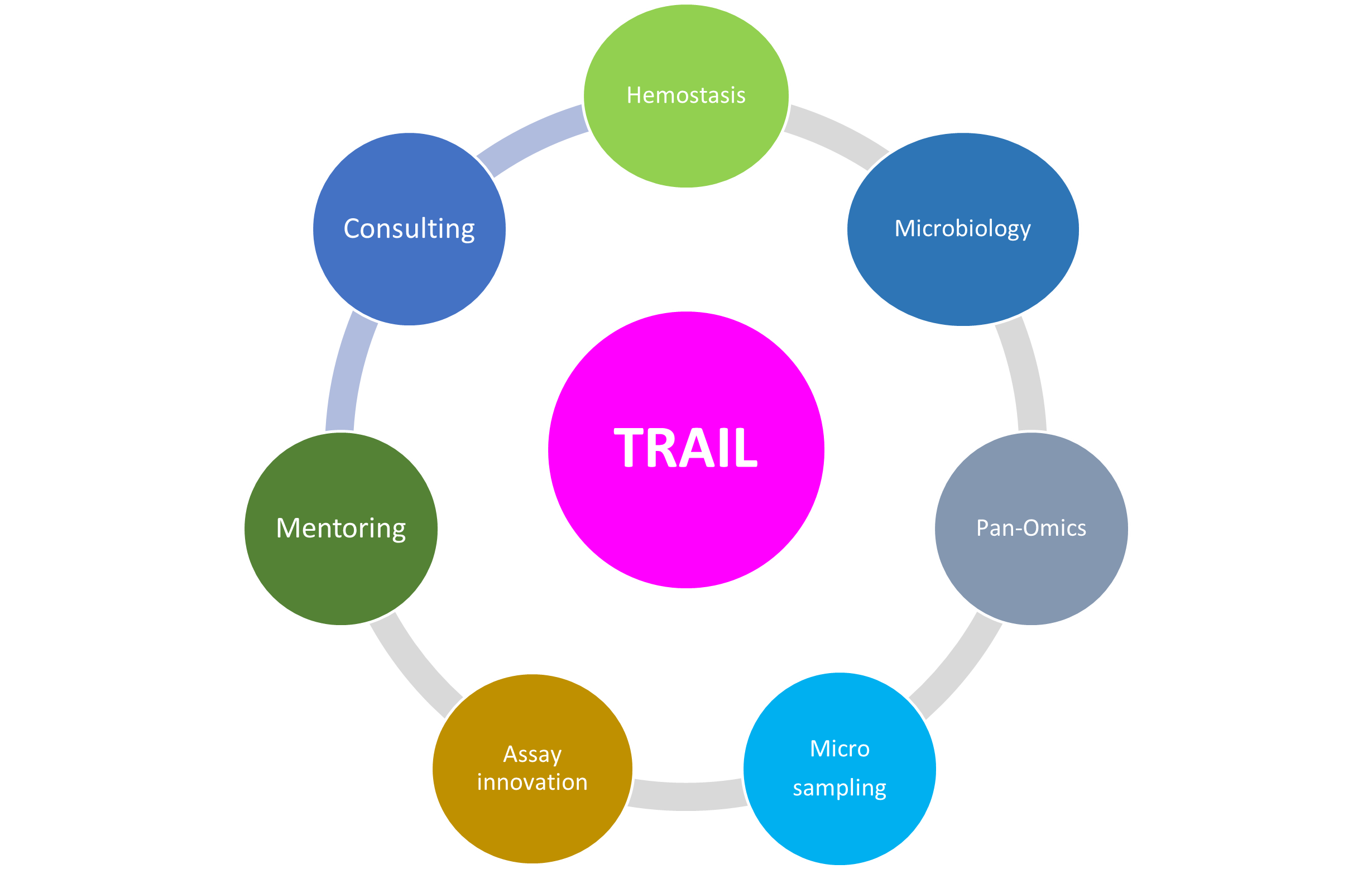
Capabilities listed by research area and/or methodological approach
| Research | Area Capabilities |
| Hemostasis |
|
| Proteomics |
|
| Whole Genome Sequencing | Promethion P2 Solo - Long read sequencing |
| Microbiology |
|
| Project Support / Specimen Management |
|
We collaborate with National and International industry partners, including global leaders in laboratory diagnostics, as well as pharma.
We are committed to mentoring and education of the next generation/s of translational researchers in the form of summer interns, and Clinical and Translational Research Training Track (CTRT) mentees.
Research Education & Training | Johns Hopkins Medicine
CTRT mentees
- Kayla Delaney, MD, 3rd year Pediatric Resident, Johns Hopkins All Children's Hospital
- Michelle Hojnicki, DO, 3rd year Maternal Fetal and Neonatal Institute (MFNI) Fellow, Johns Hopkins All Children's Hospital
- Alexa Roberts, MD, 2nd year Pediatric Critical Care Medicine (PCCM) Fellow, Johns Hopkins All Children's Hospital
- Meghan Roddy, PharmD, Clinical Pharmacist, Pediatric Intensive Care Unit, Johns Hopkins All Children's Hospital
- Brett Russi, MD, 3rd year PCCM Fellow, Johns Hopkins All Children's Hospital
- Rita Wyrebek, MD, MFNI Faculty, St Louis University, Missouri
Interns
- Juliana Hodge (current)
- Lauren White (past)
- Matthew Danielson (past)
Funding
- NIH
- Johns Hopkins All Children’s Foundation
Contact