The Johns Hopkins Department of Physical Medicine and Rehabilitation (PM&R), in collaboration with the Johns Hopkins University Applied Physics Laboratory and the Department of Neurology and Neurosurgery, has been awarded a grant by the Defense Advanced Research Projects Agency (DARPA) to conduct a clinical trial focused on recording and stimulating the brain of a person with quadriplegia.
This study is registered under ClinicalTrials.gov under the registration number NCT03161067.
Controlling Assistive Devices
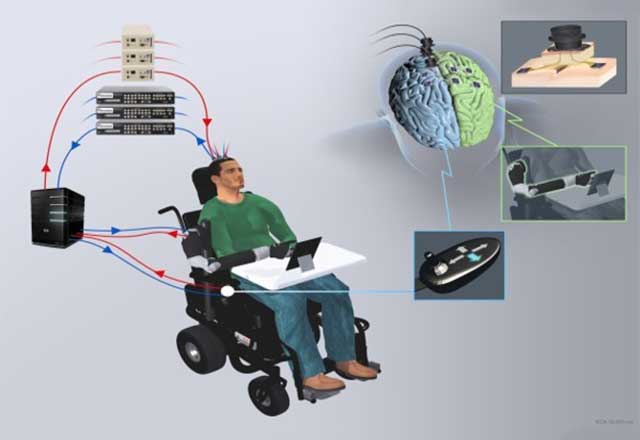
The implanted MEAs allow a computer to read the signals coming from the participant’s motor cortex. These signals convey the intention of movement. The computer then decodes and transmits these signals to the robotic arm, allowing the participant to control its movements similar to if it were the real arm.
The research team is investigating uses for the MEAs beyond controlling prosthetics. For example, the participant uses neurosignals to control a cursor on a computer or provides instructions to a smart home setup.
Additionally, we are testing whether the participant will be able to control two things at once using only neurosignals, such as controlling a wheelchair and a robotic arm at the same time.
Perceiving Physical Stimuli
While controlling a prosthetic limb is groundbreaking for someone with paralysis, it only solves half the problem. Without sensation, the participant would be controlling an arm that is essentially numb. Without sensory feedback, it is difficult to use the correct amount of pressure to pick up a cup, for example. Sensation also allows the participant to know where the arm is without having to look at it.
The goal behind implanting MEAs into the participant’s sensory cortex is to simulate sensation so that when the prosthetic arm is touched, the participant would be able to feel that touch and pinpoint its location. This ability will make robotic limbs much easier to use.
The research team wants to take perception beyond the scope of prosthetics as well. The goal is to use sensory cues to provide feedback for certain actions, such as a tap on the pinky indicating that a smart home system locked the front door.
Highlights of Our Study
Bilateral Decoding
Our team was the first to implant MEAs in both hemispheres of the brain. With this bilateral implantation, we aim to allow the participant to control right and left robotic arms independently and perceive touch on both hands simultaneously, differentiating between them.
Bilateral Sensory Stimulation
Our study was the first to deliver artificial touch sensations to the fingertips using MEAs, on both hands at the same time. We’re working to make those sensations more natural.
Research Insights
Sensory Feedback through Brain Stimulation
Characteristics and Stability of Sensorimotor Activity
The Research Team
This study is a close collaboration between several Johns Hopkins groups.
Physical Medicine and Rehabilitation
Johns Hopkins Applied Physics Lab
Francesco Tenore, M.S.E., Ph.D.
Matthew Fifer, Ph.D.
Brock Wester, Ph.D.
Breanne Christie, Ph.D.
Luke Osborn, Ph.D.
Neurology and Neurosurgery
William (Stan) Anderson, M.A., M.D., Ph.D.
Nathan Crone, M.D.
Recent Publications
- Perceived timing of cutaneous vibration and intracortical microstimulation of human somatosensory cortex
- Intracortical microstimulation of somatosensory cortex enables object identification through perceived sensations
- Novel intraoperative online functional mapping of somatosensory finger representations for targeted stimulating electrode placement: technical note