Research News Tip Sheet: Story Ideas From Johns Hopkins
04/09/2020
During the COVID-19 pandemic, Johns Hopkins Medicine Media Relations is focused on disseminating current, accurate and useful information to the public via the media. As part of that effort, we are distributing our “COVID-19 Tip Sheet: Story Ideas from Johns Hopkins” every Tuesday, throughout the duration of the outbreak.
We also want you to continue having access to the latest Johns Hopkins Medicine research achievements and clinical advances, so we are issuing a second tip sheet every Thursday, covering topics not related to COVID-19 or the SARS-CoV-2 virus.
Stories associated with journal publications provide a link to the paper. Interviews with the researchers featured may be arranged by contacting the media representatives listed.
WOMEN REPORT FEELING EFFECTS OF CANNABIS CHEMICAL CBD WHEN VAPED
Media Contact: Vanessa McMains, Ph.D.
The cannabis plant contains over 100 different cannabinoids, including ones such as delta-9-tetrahydrocannabinol (THC), which can produce a ”high” feeling and cause impaired cognitive function, or cannabidiol (CBD) which is touted for its medical and wellness properties. CBD is perceived by the public to not produce a psychoactive “high” or impairing effects the same way THC does. Researchers at Johns Hopkins Medicine have just completed a study suggesting that pure CBD does produce subjective drug effects if vaped in large doses, especially among women, but that these effects are different from those produced by THC.
“The participants had trouble describing the drug effect but many said they definitely felt different, and women reported feeling stronger effects than men overall,” says postdoctoral fellow Tory Spindle, Ph.D., a researcher in the Behavioral Pharmacology Research Unit at the Johns Hopkins Bayview Medical Center.
Drug effects were not felt by study participants when CBD was swallowed. And, the researchers confirmed with testing that CBD didn’t slow motor coordination or responsiveness when it was swallowed or vaped.
“When you think about the workplace and driver safety, typical drug effects seem to impair a person’s reaction time, which can lead to mistakes and accidents,” says Ryan Vandrey, Ph.D., associate professor of psychiatry and behavioral sciences at the Johns Hopkins University School of Medicine. “But we didn’t see that in our study with CBD.”
In their study, published on April 1, 2020, in the journal Drug and Alcohol Dependence, the researchers note that their findings suggest there is still much unknown about the various chemical components in cannabis.
COLORECTAL CANCER SCREENING AT AGE 40 MAY BE COST-EFFECTIVE
Media Contact: Valerie Mehl
It may be cost-effective to begin screening for colorectal cancer starting at age 40, instead of the typical age of 50, according to researchers at the Johns Hopkins Kimmel Cancer Center.
The medical literature indicates that the incidence of colorectal cancer is increasing among patients under the age of 50, says senior author Nilofer Azad, M.D., associate professor of oncology and co-director of the Colorectal Cancer Research Center of Excellence and the Cancer Genetics and Epigenetics Program at the Johns Hopkins Kimmel Cancer Center. For example, a 2017 study in the Journal of the National Cancer Institute reported that 30% of rectal cancers in the United States are now diagnosed in patients under age 55. That’s a trend oncologists report observing in their clinics, Azad says.
Azad and colleagues developed a computerized model to assess the cost-effectiveness of screening average-risk 40-year-olds using colonoscopy, flexible sigmoidoscopy and three types of stool tests: a fecal occult blood test, a fecal immunochemical test and fecal immunochemical DNA testing. They weighed factors, such as cost for screening and quality of life years added.
Study results, published online on Jan. 27, 2020, in the journal Preventive Medicine, found colorectal cancer screening at age 40 to be cost-effective using each of these modalities when compared with the standard of care of no screening until age 50. Flexible sigmoidoscopy provided the most favorable monetary benefit ($3,284 per person). Earlier screening suggested a potential benefit of 2.1 million quality of life years and savings of $21 billion over the lifetime of the age group.
“Our study does not definitively say that people aged 40 and up should be screened, but it does suggest that organizations that set national screening guidelines should be exploring our model and others to determine whether age 40 is a better target,” Azad says.
Indeed, last year, the American Cancer Society lowered their age for baseline colorectal cancer screening from 50 to 45.
RESEARCHERS MIMIC HUMAN LUNG ARTERIES FOR PULMONARY HYPERTENSION STUDIES
Media Contact: Michael E. Newman
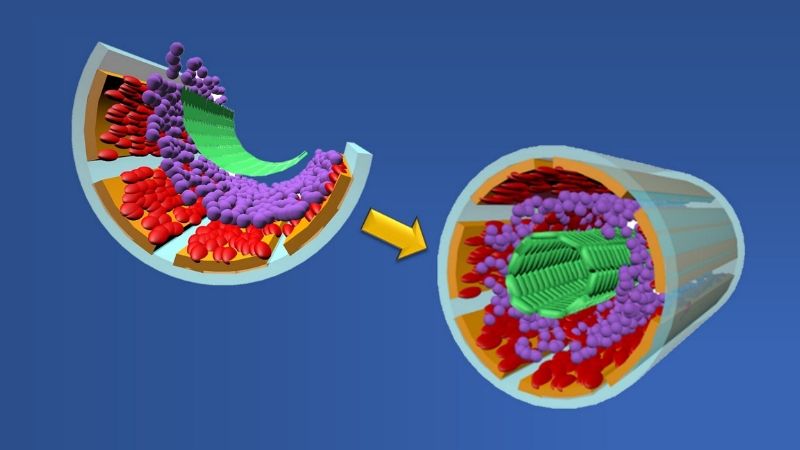
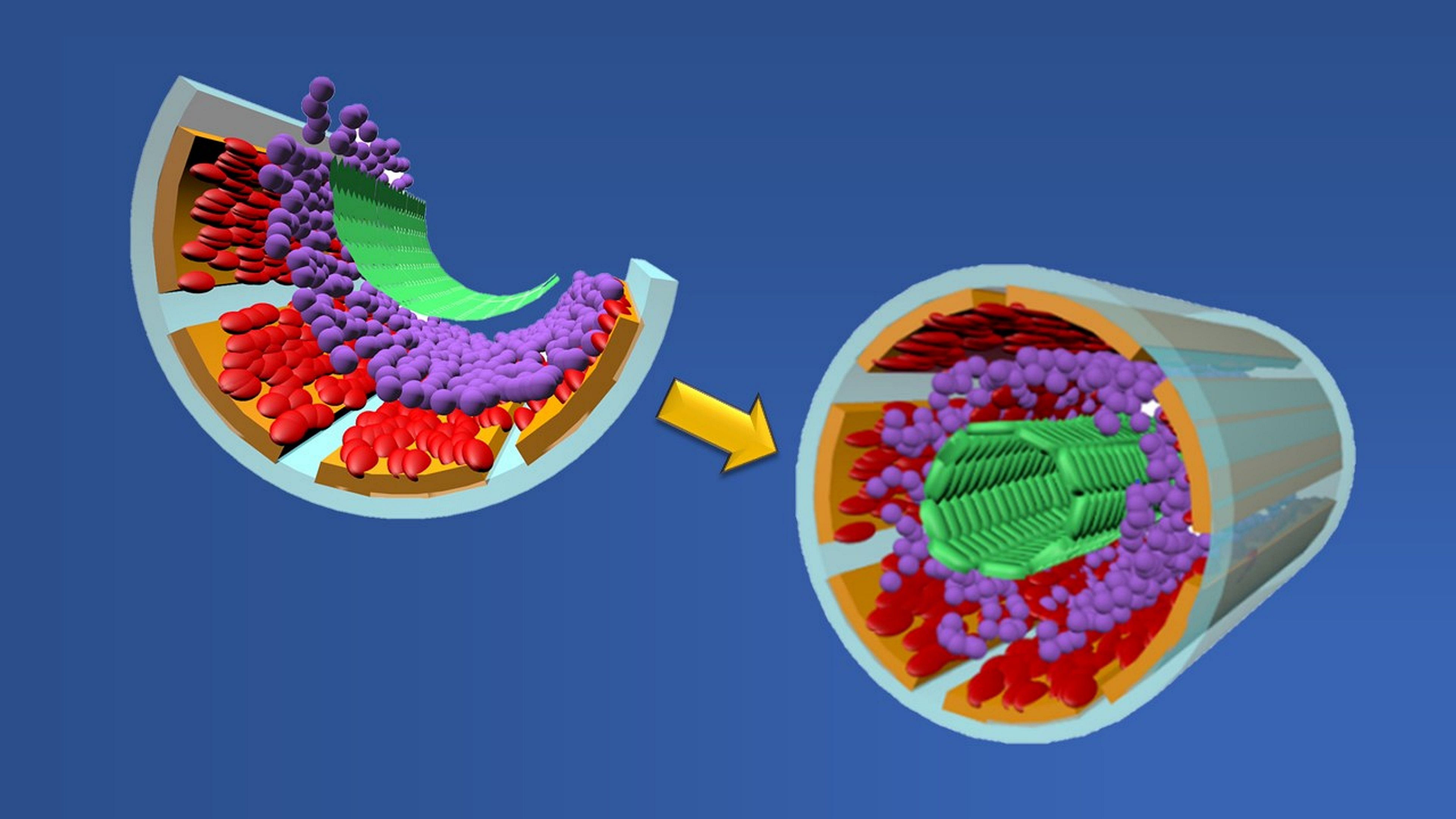 |
Graphic showing formation of a biomimetic small muscular pulmonary artery. The process involves stacking two cell types (red and green layers), two binding proteins (orange and purple layers) and a chemical coating (gray layer) atop a microchip wafer. When the wafer is removed, the layers fold upon themselves to form a tubular-shaped working copy of a blood vessel. Credit: Graphic created by M.E. Newman, Johns Hopkins Medicine, using artwork by A. Hou, The Johns Hopkins University School of Engineering |
In patients with pulmonary artery hypertension, or PAH, there is a narrowing, blocking or loss of function of small muscular pulmonary arteries, the tiny vessels that help carry blood from the heart to the lungs to exchange carbon dioxide for oxygen. Reduced function of these channels — the smallest in the body — causes the heart to pump harder than normal, raising the lung arterial pressure to dangerous levels.
To better understand the structural and physical changes that occur during PAH, a team of researchers at Johns Hopkins Medicine and The Johns Hopkins University — led by Qianru Jin, Ph.D.; David Gracias, Ph.D.; and Lewis Romer, M.D. — has developed a scalable and mass-producible biomanufacturing method that incorporates fabrication processes for computer microchips to create biomimetic (“mimicking nature”) small muscular pulmonary arteries. These artificial vessels consistently duplicate the structure, physical function and microenvironment of their real-life models. They can be used in a variety of highly controlled, repeatable and real-time laboratory studies that don’t require human or animal participants.
The novel production of these lung artery mimics is described in a paper in the March 25, 2020, issue of the journal Science Advances.
To create their biomimetic vessels, the researchers first layer the four structural components of a human pulmonary artery — smooth muscle cells (that are used to move blood), fibronectin and laminin (proteins that hold cells together) and endothelial cells (that form the vessel’s inside lining) — atop a silicon wafer coated with germanium and deposits of silicon oxide and silicon dioxide. Once the germanium and wafer are removed, the remaining materials fold themselves into tube-shaped, three-dimensional structures that look and act like small pulmonary arteries.
The researchers say they expect that their biomimetic small muscular pulmonary arteries will not only advance the study of PAH but also could pave the way for drug screening for a number of diseases where microvascular function is a factor in keeping vital organs healthy.
GENE VARIANT IN NONCODING DNA LINKED TO HEART FAILURE
Media Contact: Vanessa Wasta, M.B.A.
When scientists scour the genome for disease-causing culprits, they wouldn’t ordinarily look in so-called noncoding regions, areas of repetitive DNA that do not code for proteins. Yet, that’s exactly where Johns Hopkins scientists found genomic variations in a new study of people with heart failure.
Marios Arvanitis, M.D., a postdoctoral cardiology fellow at the Johns Hopkins University School of Medicine, and the study’s first author, completed a meta-analysis of data from more than 400,000 people and discovered variations in a noncoding region of chromosome 1 that were associated with the development of clinical heart failure. He confirmed the finding in another group of more than 1 million study participants.
The research is reported in the Feb. 28, 2020, issue of the journal Nature Communications.
The scientists’ next challenge was to uncover the function of the DNA in the noncoding region where the variations were found. “This area of the genome is probably involved in regulating how genes are turned on and off, but we wanted to know which gene the code controlled,” says Alexis Battle, Ph.D., associate professor of biomedical engineering and computer science at The Johns Hopkins University and senior author of the study.
To determine the noncoding region’s function, the scientists made a 3D model of developing human cardiac cells and used computer algorithms to determine the DNA’s structure within the cells. They found that the noncoding region folds up and ‘touches’ a nearby gene called ACTN2, which makes a protein important for the structure of cardiac cells.
Then, the investigators used the CRISPR gene-cutting tool to remove the noncoding region on chromosome 1 in lab grown cardiac cells and found that gene expression of ACTN2 drops by 47% and no other genes in that region were disrupted.
“This is a strong example of a connection between noncoding and coding regions of the genome,” says Battle. “While the variations occur in a relatively small percentage of people with heart failure, our findings may point to a genetic pathway for which a treatment can be developed — not only for people with the gene variant, but perhaps others with heart failure.”
STUDY SHOWS PROMISE IN TREATMENT OF METABOLIC SYNDROME
Media Contact: Patrick Smith
Johns Hopkins pediatrics professor Subroto Chatterjee and his colleagues have engineered a compound that not only improves triglycerides, cholesterol and type 2 diabetes in mice, but also reduces body fat. The researchers say the results of the study, published online on Feb. 24, 2020, in the Journal of Biochemical and Biophysical Research Communications, could have important implications for treating people with metabolic syndrome.
Certain lipids found in cell membranes that help cells communicate with one another — known as glycosphingolipids — have long been associated with heart disease and type 2 diabetes. Chatterjee and his colleagues sought to improve on a chemical compound designed to interrupt the body’s production of these lipids, thereby improving the conditions that contribute to metabolic syndrome.
Among the problems researchers have faced is that the compound is metabolized in the bloodstream too quickly to be very effective, unless given in high doses.
In their study, Chatterjee and colleagues encapsulated the compound in a biopolymer — a special material designed to slow down metabolism and increase its ability to inhibit the production of the glycosphingolipid at very low doses.
Metabolic syndrome conditions include high blood pressure, elevated blood sugar, high levels of LDL (“bad cholesterol”) and triglycerides and low levels of HDL (“good cholesterol”), as well as excess body fat around the waist, each of which increases a person’s risk of diabetes, stroke and heart or blood vessel disease. As many as one-third of U.S. adults has at least one of the components of metabolic syndrome.
The Johns Hopkins team administered the compound to type 2 diabetic mice over the course of six weeks. They observed marked improvements in the animals’ weight, cholesterol and diabetes, as well as in their arterial health.
“Although we proposed a relationship between glycosphingolipids, high cholesterol and hardened arteries decades ago,” Chatterjee explains, “we’ve only recently had access to the compound that inhibits the synthesis of these particular lipids.”
NEW GUIDELINES PROMOTE BETTER DIAGNOSIS AND MANAGEMENT OF PREDIABETES IN YOUTH
Media Contact: Michael E. Newman
According to the U.S. Centers for Disease Control and Prevention, prediabetes is a serious health condition in which blood sugar levels are higher than normal, but not high enough to meet the threshold for type 2 diabetes. If diagnosed early, experts say, lifestyle changes such as weight loss and regular exercise can prevent or delay the development of type 2 diabetes and the increased risks it poses for future heart disease, stroke, kidney failure and nerve damage.
Unfortunately, a 2019 Johns Hopkins Medicine national survey of primary care physicians revealed significant gaps in the group’s overall knowledge of risk factors, diagnostic criteria and recommended management/prevention practices for prediabetes. These gaps likely contribute to doctors underscreening for and missing diagnoses of prediabetes, and in turn, not intervening or referring patients to available type 2 diabetes prevention programs.
“The problem of inadequate diagnosis, management and intervention of prediabetes is even greater for children and adolescents because the way these three are defined for youth is largely based on adult data, even though type 2 diabetes can manifest itself very differently in both,” says Sheela Magge, M.D., M.S.C.E., associate professor of pediatrics and director of the Division of Pediatric Endocrinology and Diabetes at the Johns Hopkins University School of Medicine.
To increase awareness and better understanding about prediabetes and type 2 diabetes in overweight and obese children and adolescents — and to provide guidelines for screening, diagnosing and managing those with the conditions — five pediatric endocrinologists from the Pediatric Endocrine Society formed an expert working group in 2012. Magge served as chair of the team. The results of their effort are published as a review paper in the March 3, 2020, issue of the Journal of Pediatrics.
For their paper, Magge and her colleagues first reviewed the available data from prediabetes clinical trials in both adults and youth. Using this research, they constructed a flow chart detailing a screening, diagnosis, treatment and management plan tailored for a younger population.
For example, based on the results of either a fasting blood sugar or hemoglobin A1c test, the chart provides four distinct courses of action for the pediatrician to follow. The chosen path can recommend options such as lifestyle modifications, more extensive testing for better diagnosis, referrals to specialists and follow-up care — all designed specifically for children and adolescents, rather than adapted from adult prediabetes and type 2 diabetes management plans.
“Our flow chart and strategies are suggested tools for the pediatric primary care provider, who may or may not have access to subspecialists such as pediatric endocrinologists,” Magge says. “As there are no conclusive data published, we hope that our paper will provide some needed guidance to pediatric clinicians caring for overweight and obese youth.”
