Patient Story
Novel Stem Cell Transplant: Jocelyn's Story
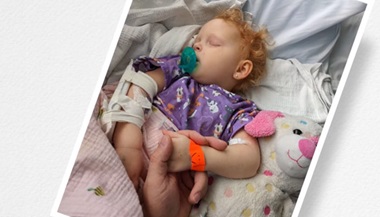
Jocelyn remembers the early wakeup calls for the first of three daily rounds of medication. She remembers when she had surgery to remove part of her lung. She remembers another surgery to remove part of her colon.
While other kids had sleepovers and frolicked on the playground, Jocelyn carefully avoided smoke, several foods and running or jumping. She wore a mask to avoid lung infections, and she hid from the sun.
“It was kind of hard,” says Jocelyn, 14. “I wasn’t allowed to do much stuff that other kids could.”
Jocelyn was born with chronic granulomatous disease (CGD), a primary immunodeficiency disorder (PIDD). It impacted her social life and often forced her to do school at home in Land O’Lakes, Florida.
The outlook for Jocelyn was bleak until she found Johns Hopkins All Children’s Hospital in St. Petersburg as it developed the Center for Cell and Gene Therapy for Non-Malignant Conditions (CCGNC).
CGD: A Rare Genetic Disorder
CGD is a rare genetic condition that affects about 1 in 200,000 people. In these cases, white blood cells — phagocytes — are unable to kill certain types of bacteria and fungi. People with CGD are highly susceptible to frequent and sometimes life-threatening bacterial and fungal infections, and need to take daily medications to stay healthy. Some patients also develop chronic inflammation involving their lungs, liver and intestines.
CGD is an inherited genetic disorder, which means that it is passed from parents to their children. The most common form of CGD is X-linked (“sex-linked”) and the other types are autosomal recessive. Females have two X chromosomes and males have an X and a Y. X-linked CGD is caused by mutation in the X chromosome, so males are exclusively affected and females are usually “carriers” since they have one X chromosome without the defect. Jocelyn has a different form of CGD, one known as an autosomal recessive form that affects both males and females equally.
Jocelyn was diagnosed with CGD as an infant and had a variety of related health issues growing up, including the lung and colon procedures.
“What she developed was a rare, unique and very severe form of the disease,” says Deepak Chellapandian, M.D., medical director of the Center for Cell and Gene Therapy for Non-Malignant Conditions. “She developed inflammation of her lungs that caused deterioration of her lung functions and breathing issues, and then she had inflammation of her colon that presented with repeated episodes of bleeding from her rectum, ultimately requiring removal of a large piece of her colon.”
CGD commonly is treated with antibacterial and antifungal medications, in some cases with anti-inflammatory medications, but as Jocelyn’s case became more severe, she became a candidate for a bone marrow transplant.
But that requires a matching donor.
Seeking a Match
Matches for marrow transplantation are based on a patient’s human leukocyte antigen (HLA), which is a protein — known as a marker — found on most cells in your body and is responsible for regulation of the immune system.
Successful transplants traditionally have depended on a close HLA match between patient and donor, particularly in certain markers.
As Jocelyn’s doctors looked for a matching donor, they kept striking out. The registry is largely represented by Caucasian donors who often are not a close match with minority patients, such as Jocelyn, who is Hispanic. Chellapandian says they find a close match for a Hispanic patient only 15 to 20 percent of the time.
“Unfortunately, the donor registry didn't reveal any good donors,” Chellapandian says. “She was managed with supportive care for several years, meaning giving her antibiotics for infections and steroids and immunosuppressive medications to control inflammation.”
A New Approach
Johns Hopkins Medicine pioneered a technique to successfully do stem cell transplants with “half-matched” donors. A person inherits half of his or her chromosomes from each parent, so the parents are a half-match and often siblings are as well. This expanded the pool of potential donors for many patients. The technique has been successful for certain leukemia and other blood disorders, but encountered some challenges with conditions such as CGD.
“We started doing half-matched transplants a long time ago,” Chellapandian says, “however, these transplantations historically have been very complex to perform, and in some cases, lead to a lot of unintended complications.”
Chellapandian, who specializes in bone marrow transplant for rare diseases such as CGD, developed a protocol for using the half-match transplant — known as haploidentical bone marrow transplant — for conditions such as Jocelyn’s with fewer complications. It is the first of its kind in Florida.
Jocelyn’s brother, Miguel, served as her half-matched donor in March 2021. Chellapandian’s team collected stem cells from him and then used a graft manipulation technique — specifically TCR alpha beta T cell depletion — a kind of cell filtration process performed outside the body using a specialized device known as CliniMACS®. This would eliminate the active immune cells known to cause complications such as graft versus host disease and preserve the “essential” cells that would help the recipient’s body accept the donor’s stem cells and fight against infections.
“Fortunately, she sailed through the transplant procedure without facing any major complications,” Chellapandian says. “She is now eight months from her transplant, her body has accepted her brother’s graft, and she is finally cured of her CGD.”
Jocelyn, who no longer has the faulty white blood cells from her CGD, is one of three successful transplants using Chellapandian’s new protocol.
“This protocol would benefit the children with rare primary immunodeficiency and hematological disorders that have been waiting on the wait list for transplant for several years due to non-availability of a matched donor,” Chellapandian says.
As a side benefit, each of the three patients who have had successful transplants under Chellapandian’s protocol have been from ethnic minority groups who traditionally have trouble finding 100 percent donor matches.
When I really knew it was going to happen was when the doctor said I no longer had the condition. It made my life better. Now I can live life without worrying too much.
Jocelyn
Thriving
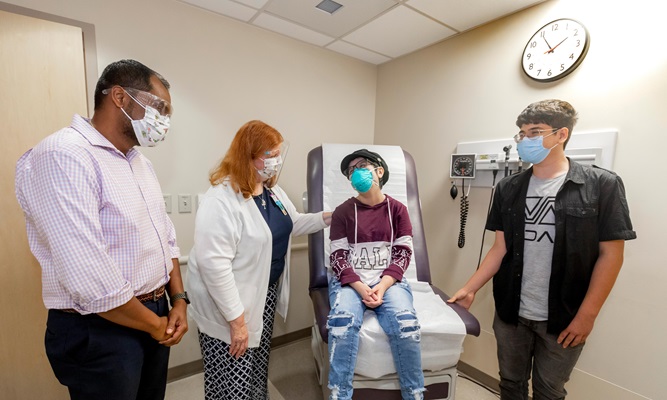
Jocelyn is grateful to Miguel, who turned 16 in July, and the team at Johns Hopkins All Children’s.
“They treated me well,” she says. “If I was scared of a shot or a CT scan, they would talk me through the process and help me understand. They were really helpful.”
Jocelyn has ditched the three rounds of daily medications. Now, she just takes a daily dose of vitamin D and gets a shot every two weeks, which she is expected to come off soon. She still sometimes wears a mask, but that is because of COVID-19 rather than CGD.
Her life more closely resembles that of other kids.
“When I really knew it was going to happen was when the doctor said I no longer had the condition,” Jocelyn says. “It made my life better. Now I can live life without worrying too much.”