Our Transplant Immunology Research
Explore Our Ongoing Research
Transplant Immunology
The goal of the Transplant Immunology section of the VCA Lab is to develop innovative and effective strategies of modulation of alloreactivity and autoreactivity, ultimately promoting lasting immune tolerance.
We believe that great advances can be made by juxtaposing the results of investigations in tangential areas, hence the additional focus of the lab on autoreactivity, type 1 diabetes in particular. Moreover, to bring a fresh perspective to the field, the advanced skillset of rodent models of skin, heart, and the unique model of hind limb transplantation that are implemented in the lab is paired with multidisciplinary approaches that range from bio-engineering and biomimetic approaches of drug delivery (for tailored and selective immune-modulation) to advance imaging approaches and mathematical modeling of the dynamics of transplant rejection.
Our overall goal is to drive the translation of different strategies of immunomodulation into successful clinical application and provide different treatment options for the minimization or complete elimination of immunosuppression in transplanted patients. We believe this will be possible via the continuous refinement of the strategies already under investigation as well as by implementing novel investigative technologies in a collaborative fashion.
Ongoing Research
Targeted modulaton of immune reactivity via biomaterials
Primary PI: Giorgio Raimondi
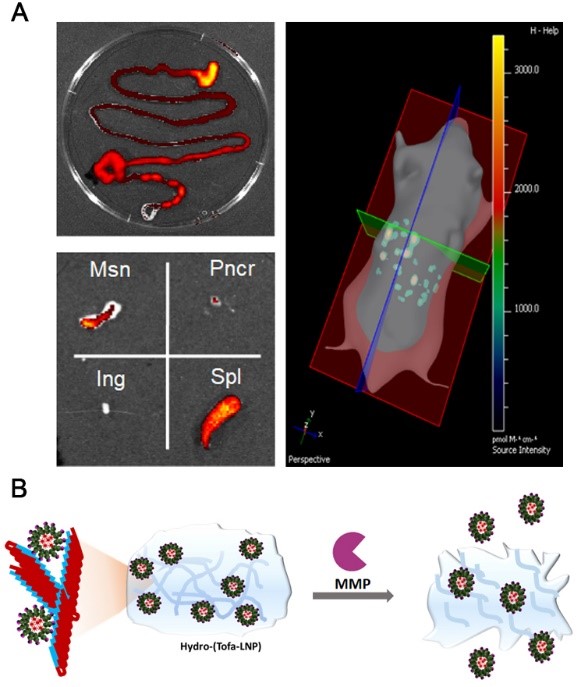 A, Representative in vivo distribution of fluorescent lipid nanoparticles following oral gavage. B, Design of an inflammation responsive hydrogel. Click to enlarge.
A, Representative in vivo distribution of fluorescent lipid nanoparticles following oral gavage. B, Design of an inflammation responsive hydrogel. Click to enlarge.Our team has a long history of collaborations with biomaterial scientists in multidisciplinary projects investigating the potential of biodegradable microparticles as a means for in vivo localized and controlled drug delivery to finely modulate immune reactivity. Ranging from modulating the maturation process of antigen presenting cells to promoting an in vivo gradient for the attraction and induction of regulatory T cells, these projects have been very successful. This approach remains an integral backbone of the research performed in the VCA lab and established novel and exciting collaborations. Via an NIH-funded R21, we have explored the potential of lipid nanoparticles in immunomodulation (with Dr. Julia Patrone, Applied Physics Laboratories). These particles have unique properties that render them one of the few formulations with potential of targeting lymphoid tissues after oral administration. Importantly, this is a very desirable clinical strategy for a broadly applicable therapeutic platform. This project evolved into a JDRF-funded strategic research agreement for the development of an antigen specific immunotherapy for the prevention of type 1 diabetes via selective targeting of localized inflammation.
In parallel, we established a collaboration with the group of Dr. Joel Schneider (NCI), whom pioneered the concept of “injectable hydrogel” for localized drug delivery. Our work is now funded by a multi-PI DoD sponsored grant to develop a hydrogel for the localized release of anti-inflammatory drugs that is also tailored to the strength of the immune response to a composite allotransplant. This work encompasses the inclusion of lipid nanoparticles (with Dr. Patrone), for the execution of a “Russian Dolls” strategy that would selectively target antigen presenting cells involved in the initiation of the rejection response. Such an approach would maximize the efficacy of currently investigated tolerogenic approaches, while minimizing any systemic impact.
Elucidation of the interplay between cytokines in the modulation of immune reactivity
Primary PI: Giorgio Raimondi
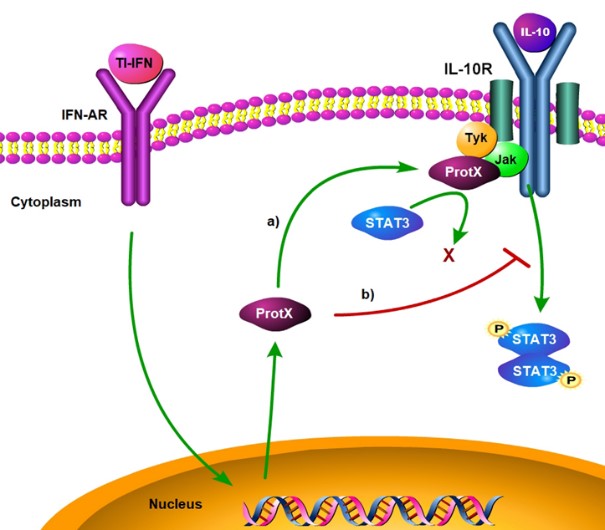 Proposed model of cross-regulation between type 1 interferons (TI-IFN) and interleukin-10 (IL-10) signaling. Click to enlarge.
Proposed model of cross-regulation between type 1 interferons (TI-IFN) and interleukin-10 (IL-10) signaling. Click to enlarge.We have a long-standing interest in the elucidation of the cross-regulation between pro- and anti-inflammatory cytokines in the modulation of the activity of T lymphocytes. Started as a project to identify strategies to improve the therapeutic efficacy of regulators of the immune system called regulatory T cells, this work has revealed the existence of a novel mechanism by which type 1 interferons impair the signaling ability of the suppressive cytokine interleukin-10. We have discovered this phenomenon underlies the development of type 1 diabetes and we are defining the level of contribution to the etiology of this disease. Through another strategic research agreement with JDRF, we are currently investigating the molecular underpinning of this cross-regulation to identify novel targets of interventions that would potentiate the promising efficacy of antigen specific immunotherapy. Overall, this mainstay of our research program has been a great opportunity to improve our knowledge of IL-10 biology and we envision it will remain the launching pad for identification of novel molecular mechanisms of immune regulation at work in healthy as well as in pathological conditions.
The Hybrid-Thymus Project: manipulating thymic selection to promote lasting protection of transplanted tissues
Primary PI: Giorgio Raimondi
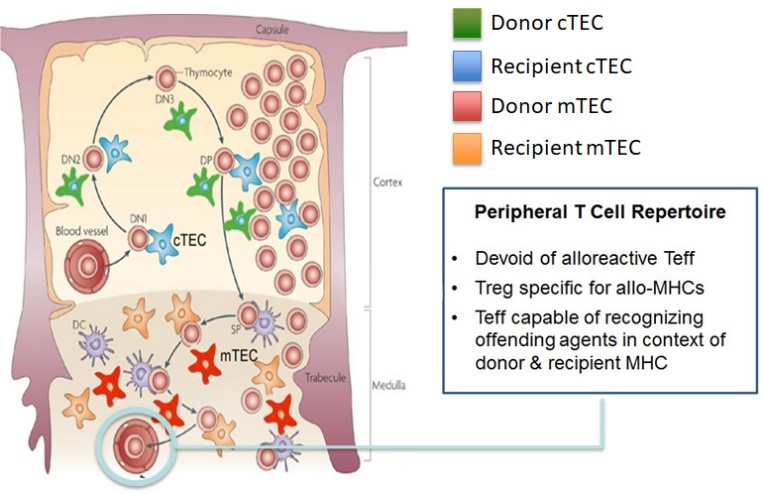 Design and function of a Hybrid Thymus. Click to enlarge.
Design and function of a Hybrid Thymus. Click to enlarge.To complement the focus on the modulation of circulating lymphocytes (peripheral tolerance), we have initiated a bold exploration of a strategy to manipulate thymic selection of T lymphocytes. In this DoD-funded discovery project, we have developed a strategy to purify donor-derived thymic epithelial cells and promote their engraftment (via intra-thymic injection) in the thymus of prospective recipients of a hind limb transplant. Many past investigations have demonstrated the key role of thymic epithelial cells in thymic selection, but only recently the improved understanding of this challenging population has revealed the potential of its manipulation to alter immune responses. We envision that the generation of a “hybrid thymus” – the coexistence of donor and recipient cells – will have a profound impact on the selection of T lymphocytes responsible for rejection as well as of regulatory T cells capable of protecting the graft. Though technically challenging, we have actively worked with our collaborator (Dr. Thomas Serwold, Joslin Diabetes Institute – Harvard Medical School) to make the generation of a hybrid thymus a reality. We are now investigating the impact of such a manipulation on the anti-donor reactivity of recipient mice. This project will add to our understanding of thymic selection and will test the validity of a strategy that could radically change how we approach tolerance induction in transplantation and beyond.
CAR-Treg therapy in VCA
Primary PI: Giorgio Raimondi
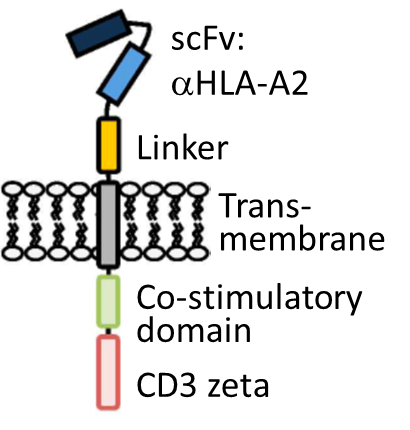 Diagram of a chimeric antigen receptor.
Diagram of a chimeric antigen receptor.Our team has a long-standing focus on harnessing one of the natural ways that the immune system uses to regulate itself: a special type of white blood cells known as T regulatory cells (or Tregs). This population has the fundamental ability to prevent inappropriate immune responses. We know from others’ and our own research that this function of Tregs can be applied in transplantation, and, at least in animal models, can promote long-term graft acceptance in the absence of long-term immunosuppressive therapy. Our collaborator, Prof. Megan Levings (University of British Columbia) has pioneered a significant improvement in how Tregs can be used therapeutically in transplantation by applying a cell engineering strategy that has led to a revolution in cancer immunotherapy. This strategy involves the creation of a custom-designed protein called a chimeric antigen receptor (CAR) which, when expressed on Tregs, makes them much more potent and specific. First-in-human clinical trials of CAR-Treg therapy in transplantation are about to begin. Our combined expertise in Treg biology, VCA immunotherapy, and CAR-Tregs positions us to make rapid progress in this highly innovative, yet clinically feasible, approach.
We hypothesize that CAR-Treg specific for an MHC Class I antigen (HLA-A2, commonly expressed by transplanted tissues) will be an effective aid in minimizing VCA rejection. Our objectives are to use mouse models of VCA (including humanized mice models) to: 1) identify the conditions that maximize the therapeutic effect of CAR-Tregs in promoting VCA long-term survival; 2) optimize the function of CARs in Tregs to promote longevity, stability and tolerance induction; and 3) demonstrate the translatability of human CAR-Treg therapy in VCA. Overall, this project aims at revealing the strengths and limitations of this technology and ultimately support a clinical trial for the application for CAR-Treg therapy in VCA.
Tolerogenic artificial Antigen Presenting Cells (TolAPC) for expansion of antigen specific Treg
Primary PI: Giorgio Raimondi
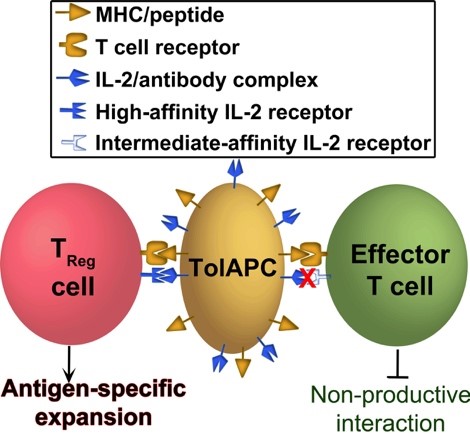 TolAPCs are designed to stimulate TReg but not effector T cells.
TolAPCs are designed to stimulate TReg but not effector T cells.Another project of our laboratory tackles the potential of Treg therapy from a different perspective, one that embraces bioengineering and the use of biomaterials. There is growing interest in the development of an off-the-shelf, non-cellular product that could stimulate the expansion and activity of protective Treg directly in the body of the patients. Currently, the most promising approach is the use of “low-dose IL-2 therapy.” However, a protective effect of IL-2 in transplantation has yet to be found, with reports indicating limited advantages. We believe this shortcoming derives from two main phenomena: 1) IL-2 promotes the nonspecific expansion of all Treg that recognize self-antigens, diluting and limiting the effect of graft-specific Treg; 2) once activated, conventional T cells become sensitive to low-dose IL-2, and are further stimulated. Therefore, we propose to test a bioengineered construct (TolAPC) that will deliver IL-2 selectively to allograft-specific Treg. To achieve this, we teamed with material scientist Dr. Jordan Green and bioengineer Dr. Jamie Spangler in an exploratory project funded by the DoD and the JDRF. We are investigating biodegradable micro- and nano-particles that are coated with: 1), an engineered version of IL-2 that is selective for Treg, together with 2) donor MHC-peptide complexes. In such a configuration, IL-2 will be preferentially directed to Treg that are specific for donor MHC-peptides, thus promoting their in vivo enrichment and function. The absence of costimulatory signals will prevent activation of pathogenic alloreactive T cells. Moreover, the use of particles will enable precise distribution of the reagents in the body, minimizing the chances of effector T cell stimulation. Because of its modular design, we believe TolAPC is a versatile platform technology that can be implemented in other pathological scenarios like type 1 diabetes and other autoimmune diseases.
Mathematical modeling of transplant rejection
Primary PI: Giorgio Raimondi
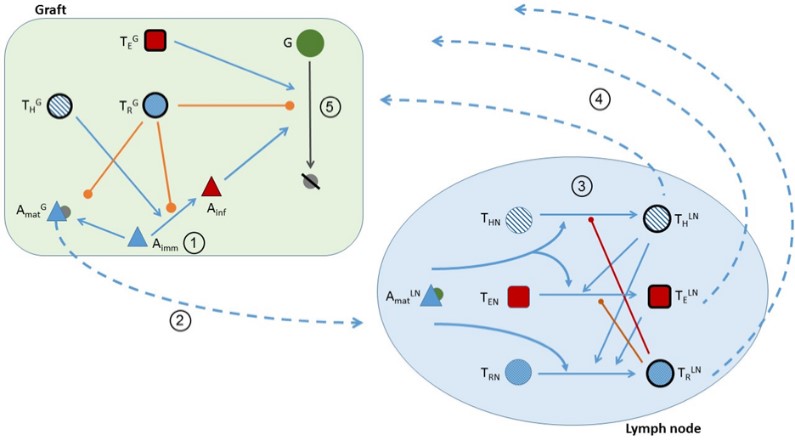 Diagram of the interactions defined in our ODE model of transplant (graft) rejection. Click to enlarge.
Diagram of the interactions defined in our ODE model of transplant (graft) rejection. Click to enlarge.A frustrating criticism of transplant research (and other fields) is that despite promising pre-clinical results, clinical implementation of immunotherapies has very often been disappointing. In addition to the perpetual re-evaluation of animal models, we believe that this disconnect between experiment and the clinic is likely due to studying rejection from a single component perspective rather than as a system. Mathematical modeling transcends investigations of individual processes by unraveling relevant mechanisms and identifying emerging properties of a complex system. Applying computational modeling to better understand transplant rejection and aiding in designing more effective interventions is an exciting opportunity. Collaborating with mathematician Dr. Julia Arciero (IUPUI, Indianapolis), we have created the first (ordinary differential equations based) comprehensive model of rejection of a mouse heart (Arciero et al, Front. Immunol 2016). Though certainly not perfect, this model enables the rapid exploration of the impact of perturbations of the system, an approach that we are currently using to optimize an immune-modulatory therapy for rejection based on the adoptive transfer of regulatory T cells. The present challenge in the clinic is to design an optimal regimen to maximize the capacity of regulatory T cells to promote transplant tolerance. Our current work pairs theoretical predictions of regulatory T cells dosing and timing (with or without immune-modulatory drugs) with novel experiments in our mouse transplant models to identify this optimal regimen.
Autologous Hematopoietic Stem Cell Transplantation to Prevent Antibody-Mediated Rejection after Vascularized Composite Allotransplantation
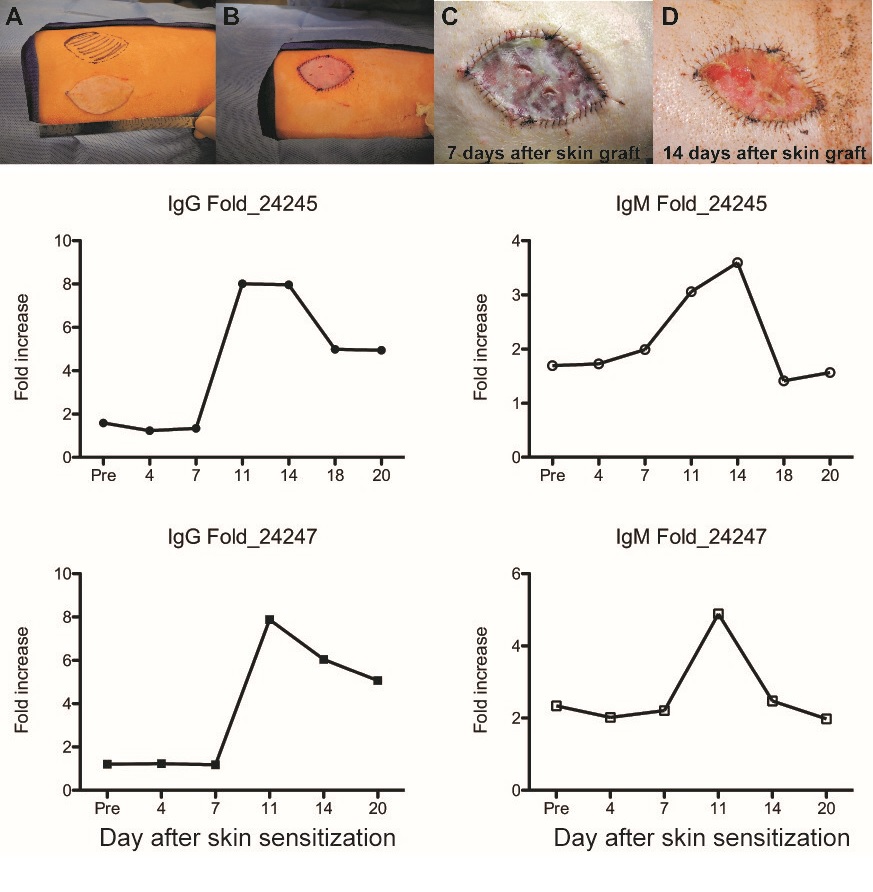 Photos of skin grafting and fold increases of donor specific antibodies formation. Click to enlarge.
Photos of skin grafting and fold increases of donor specific antibodies formation. Click to enlarge.In the field of solid organ transplantation, pre-sensitization (pre-exposure to allogeneic antigens) is known as the greatest risk factor for allograft rejection and long-term graft loss, and it frequently causes patients with donor specific antigen to be excluded as candidates for transplantation. In addition, donor specific antibody (DSA) play a crucial role in the development of antibody mediated rejection (AMR), which has emerged as the major clinical challenge and most frequent cause for renal allograft failure. In the US, about 40% of kidney transplant candidates are currently sensitized or highly sensitized. In a recent study it was shown that this number is even higher for VCA candidates. In this report, over 50% of patients with extensive third-degree burns evaluated for VCA presented with a panel-reactive antibody level >85% and were thus considered contraindicated for transplantation due to HLA-hypersensitization. In general, since sensitized transplant candidates have major disadvantages such as prolonged waiting times and increased risk of antibody mediated rejection, these patients transplantation rates are as low as 16% over an 8-year period and mortality rates approach 70%. Our team has pioneered the development and implementation of translational small and large animal models and recently performed a series of preliminary experiments that provide clear evidence of the critical importance and detrimental effects of sensitization and DSA on VCA survival. Overall, goal of this project is to comprehensively investigate the mechanisms and impact of pre-existing and de-novo DSA and AMR in VCA and to develop a clinically relevant desensitization protocol that will subsequently broaden the population of sensitized patients eligible for reconstructive transplantation.
Post-Transplantation Cyclophosphamide to Promote Immune Tolerance after Reconstructive Transplantation
In this DoD-funded collaboration with the team of Dr. Leo Luznik (Department of Oncology at the Sidney Kimmel Comprehensive Cancer Center) we are studying the impact of combining vascularized composite allotransplantation, donor bone marrow transplantation and post-transplantation high-dose cyclophosphamide (PT/Cy) treatment on immune tolerance induction. PT/Cy is unique in comparison to other immunosuppressive strategies: it takes advantage of the increased cytotoxic sensitivity of the proliferating, donor and recipient alloreactive T cell clones over resting, non-alloreactive T cells-thereby inhibiting both graft versus host effects and graft rejection.
High-Dose Post-Transplantation Cyclophosphamide to Induce Delayed Immune Tolerance After Reconstructive Transplantation
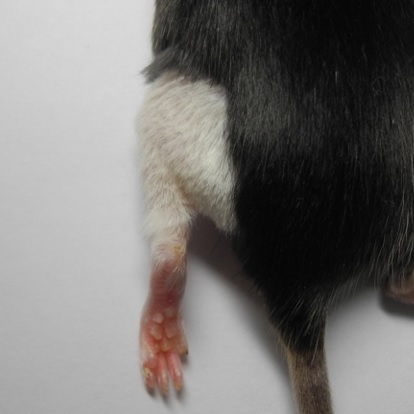 Animal after orthotopic hind limb transplantation and delayed tolerance induction on POD 1. Hind limb allograft without signs of acute or chronic rejection on POD 80. Click to enlarge.
Animal after orthotopic hind limb transplantation and delayed tolerance induction on POD 1. Hind limb allograft without signs of acute or chronic rejection on POD 80. Click to enlarge.The overall goal of this proposal is to identify mechanisms of delayed transplant tolerance induction as they specifically relate to VCA and establish a donor bone marrow (BM) and post-transplantation high-dose cyclophosphamide (PT/Cy)-based protocol for the induction of delayed tolerance with minimal or only transient immunosuppression after extremity and craniofacial transplantation. We hypothesize that a vascularized intragraft BM stromal micro-environment combined with PT/Cy treatment will promote immunoregulatory mechanisms that allow for establishing delayed tolerance and ultimately immunosuppression-free graft survival.
Using a murine model of VCA, we have identified a tolerogenic strategy that employs donor bone marrow transplantation and PT/Cy treatment (and is further facilitated by transplanting a vascularized bone marrow containing allograft). However, the ability to perform combined BMT and VCA from the same unrelated HLA-mismatched cadaveric donor requires extensive preconditioning of the recipient and thus is still limited. With this study, we are investigating the requirements to extend our established PT/Cy-based induction protocol to a “delayed” setting, which offers compelling potential to bypass this limitation in VCA. In this scenario the recipient initially undergoes VCA transplantation with conventional immunosuppression, followed by conditioning and dBMT at a later stage post transplantation.
Our preliminary results indicate that the time delay between initial hind limb transplantation and tolerance induction has a significant impact on graft survival. Additional donor bone marrow infusion promotes graft acceptance in a time depending manner. In animals with long-term allograft acceptance, flow analysis shows durable chimerism levels indicating successful engraftment of the infused donor bone marrow. Working in collaboration with Dr. Luznik (Department of Oncology at the Sidney Kimmel Comprehensive Cancer Center, Johns Hokpkins School of Medicine) to characterize mechanisms required to establish delayed tolerance and to translate and optimize efficacy of a delayed PTCy-based tolerance regimen in a clinically relevant large animal model (MGH miniature swine) for VCA.
Taming endothelial activation and sterile inflammation to rescue VCA’s from preservation injury
The current VCA donor pool is limited to the local region because of concerns over ischemia/reperfusion injury (IRI) resulting from prolonged periods of ex-vivo cold storage, an unavoidable component of organ recovery insult from cadaver sources. An oxidative stress, the hallmark of IR-tissue damage, triggers pro-inflammatory cytokine/chemokine programs, which create a deleterious milieu promoting cell death and differentiation of rejection-mediating T effector cells while hindering generation of regulatory T cells. It has been shown that minimizing IRI decreases the incidence of acute/chronic rejection and improves clinical outcomes. Thus, better protection against IR-stress should diminish innate immune-driven sterile inflammation and ameliorate host adaptive responses that otherwise act in concert to facilitate VCA failure.
This study is being carried out with collaborating partners at UCLA. The Dumont – UCLA Transplant Laboratory, is at the forefront of cutting-edge basic and translational research investigating immunological mechanisms underlying IRI in orthotopic liver transplantation (OLT). They were one of the first groups to propose that organ IRI should be regarded as an innate immunity-dominated sterile inflammation response. In parallel to “mechanistic” studies, they embarked on screening chemical compounds that rescue organ grafts from harmful IRI sequel.
Leukocyte trafficking into IR-stressed organ is a crucial multistep process, characterized by a coordinated sequence of adhesive interactions with vascular endothelial cells (EC). P-selectin (CD62) was identified as a dominant EC adhesion molecule (CAM), which supports early leukocyte/platelet tethering and rolling, ultimately leading to sequestration, activation and pro-inflammatory phenotype in the target organ. In preclinical models and in the clinical setting of liver transplantation, the UCLA group already investigated a CD62 antagonist, known as recombinant P-selectin glycoprotein ligand IgG (rPSGL-Ig), selectively blocking leukocyte adhesion and cellular homing in vivo. The UCLA team has recently constructed and tested a novel recombinant soluble form of P selectin glycoprotein ligand-1, a tandem P selectin glycoprotein ligand Ig (TSGL-Ig), with enhanced endothelial selectin binding activity as compared with the corresponding native human rPSGL-Ig.
This study will investigate if taming endothelial activation by interrupting P-selectin dependent leukocyte adhesion cascade by use of TSGL-Ig unleashes anti-oxidant/cytoprotective molecular signature and immune checkpoint activity that promotes CD4+ T cell exhaustion in the setting of murine VCA. Identifying the continuum of innate – adaptive immune phenotypes will further permit us to select, monitor and refine the practice of therapeutic interventions against IRI, eliminate the need for life-long immunosuppression, and hence improve VCA outcomes.
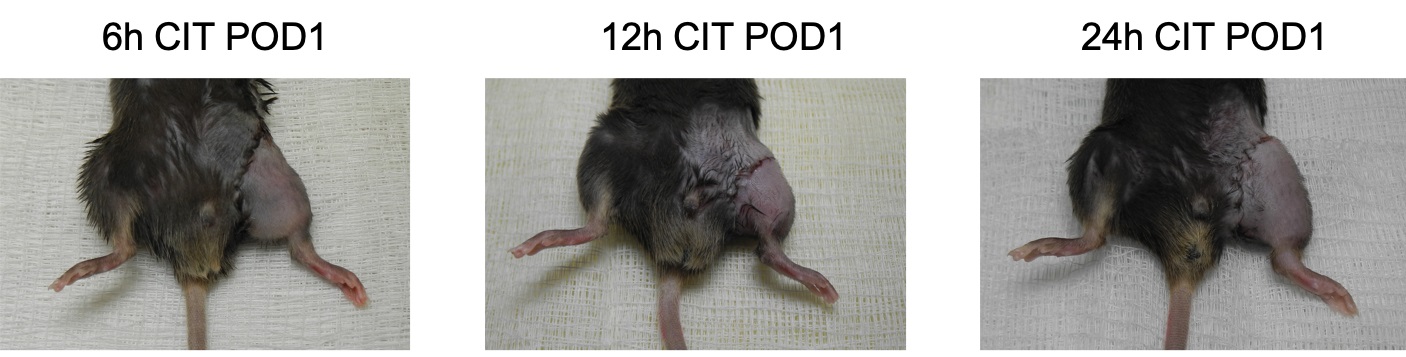 Syngeneic hind limb transplantation using conventional cold storage in HTK solution. Click to enlarge.
Syngeneic hind limb transplantation using conventional cold storage in HTK solution. Click to enlarge. Tissue Preservation and Immunogenicity
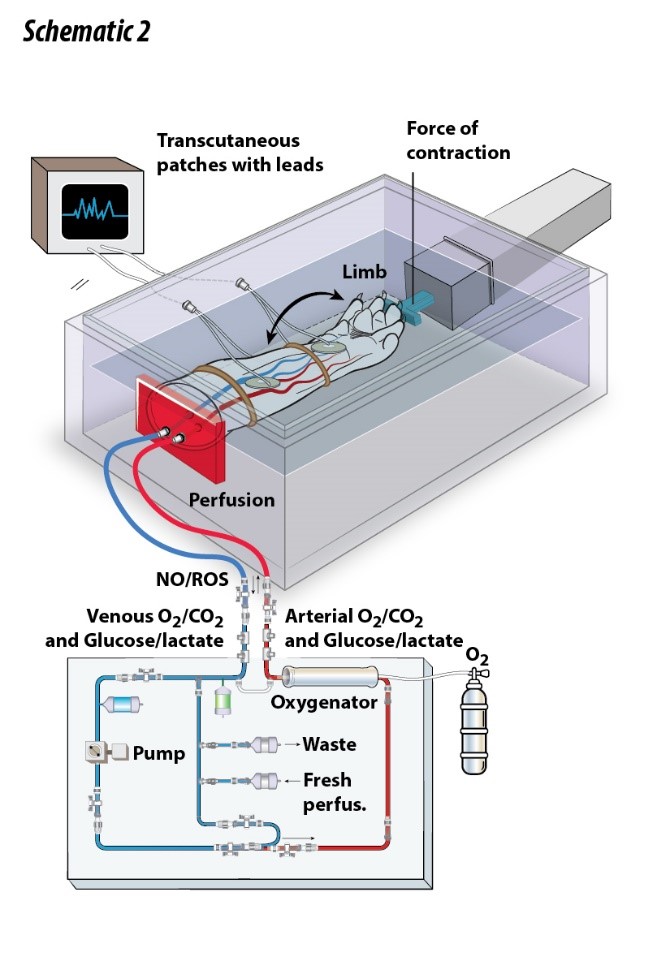
Multi-Parametric Bioreactor for Functional Preservation of Vascularized Composite Allografts
Currently, the transformational potential of vascularized composite allotransplantation (VCA) is severely limited by short preservation times (4 – 6 hours). VCA grafts suffer damage from tissue necrosis during the preservation period and increased ischemia-reperfusion injury prior to transplantation. Consequently, our laboratory believes that organ preservation can play a major role in improving the clinical outcome of transplantation. In the last decade, extracorporeal perfusion systems have been developed for solid-organ transplants (e.g. liver). Yet, there is a need for novel technologies capable of preserving the unique, functional capabilities of vascularized composite allografts. Via a Department of Defense (DoD)-funded multi-PI grant, we have established a collaboration with Dr. Warren Grayson (Johns Hopkins University, Department of Biomedical Engineering) to develop a novel, customized multi-parametric system capable of extending the preservation time and function of vascularized composite allografts. Our laboratory has pioneered two rodent models of VCA that facilitate the measurement of functional outcomes of composite allografts after transplantation. Using these models, we are currently testing several bioreactor model systems that not only minimize composite tissue necrosis, but also have been customized to provide real-time monitoring of tissue viability prior to transplantation. Such an approach would maximize the efficacy and clinical applicability of VCA.
Nerve Research
Projects
- Non-Invasive Immune Monitoring To Improve Outcomes in Composite Tissue Transplantation (Joanna)
- Long Term Banking Of Vascularized Composite Grafts Using Ice-Free Cryopreservation by Vitrification and Nano-Warming Technologies (Samuel)
- Preclinical Development of a Novel Small Molecule T Cell Metabolism Inhibitor To Prevent Rejection And Enable Calcineurin Inhibitor-Free Allograft Survival In VCA (Joanna)