Dual Immunotherapy Regimen Delays Cancer Progression in Patients with Advanced Melanoma
06/02/2021
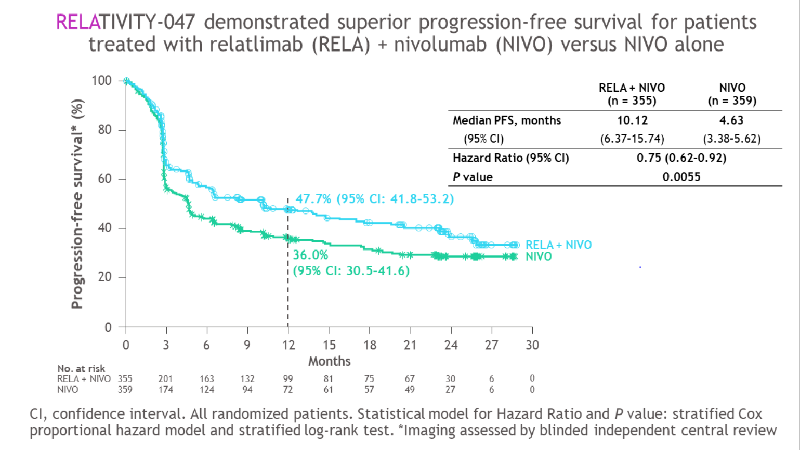
A treatment regimen for patients with advanced melanoma that combines the immunotherapy agents relatlimab (anti-LAG-3) and nivolumab (anti-PD-1) delayed time to cancer progression significantly more than nivolumab alone, according to results of a study to be presented June 6 at the 2021 American Society of Clinical Oncology (ASCO) annual meeting.
Lead study author Evan Lipson, M.D., an associate professor of oncology at the Johns Hopkins Kimmel Cancer Center and Bloomberg~Kimmel Institute for Cancer Immunotherapy, will present the findings and is available for media interviews.
The study is the first phase III randomized clinical trial to demonstrate a clinical benefit from blocking the LAG-3 immune checkpoint.
“Our results demonstrate that combination therapy with relatlimab and nivolumab is a potential new treatment option for patients with previously untreated melanoma that cannot be removed by surgery. This is the first phase III study to confirm that targeting the LAG-3 immune checkpoint is a beneficial therapeutic strategy for patients with cancer. Our findings establish the LAG-3 pathway as the third immune checkpoint pathway in history, after CTLA-4 and PD-1, for which blockade has clinical benefit,” says Lipson.
In a global, multicenter clinical trial called RELATIVITY-047, 714 patients with previously untreated, inoperable melanoma — an aggressive type of skin cancer — were randomized to receive either relatlimab and nivolumab or nivolumab alone. The median time to disease progression was 10.1 months among patients who received the combination treatment, significantly longer than the 4.6 months seen among those who received nivolumab alone. Progression-free survival (the length of time that cancer does not worsen) at one year was 47.7% for patients receiving the combination treatment, compared with 36.0% for those receiving nivolumab alone.
Checkpoint inhibitor immunotherapy works by blocking specific proteins on the surface of cells that help cancer evade the body’s immune system. Blocking these checkpoints helps the immune system fight and eliminate cancer. Nivolumab acts on a protein called PD-1 and is FDA-approved for treating melanoma and several other cancer types. Relatlimab blocks the signaling of an inhibitory protein called LAG-3 displayed on immune system T cells, reinvigorating their anti-tumor activity. The anti-tumor effects of LAG-3 blockade were originally co-discovered by scientists at the Bloomberg~Kimmel Institute.
In general, the adverse events associated with nivolumab and relatlimab were manageable and reflected the safety profile typically seen with immune checkpoint inhibitors. Grade 3 and 4 treatment-related adverse events such as fatigue or joint pains were more common among patients receiving the combination therapy versus nivolumab alone (18.9% versus 9.7%). Treatment-related adverse events led to therapy discontinuation in 14.6% of patients receiving the combination therapy versus 6.7% of those receiving single-agent therapy. Treatment-related deaths were rare: three among patients who received the dual regimen and two with nivolumab alone.
The trial was sponsored by Bristol-Myers Squibb.
Funding, materials, and drugs for the study described in this press release were provided by Bristol-Myers Squibb Co. Dr. Evan Lipson, Dr. William Sharfman, and Dr. Suzanne Topalian are paid consultants or advisory board members to Bristol-Myers Squibb Co. This arrangement has been reviewed and approved by the Johns Hopkins University in accordance with its conflict of interest policies.
