Alcohol Relapse Rate Among Liver Transplant Recipients Identical Whether or Not There is A 6-Month Wait Before Transplant
04/25/2019
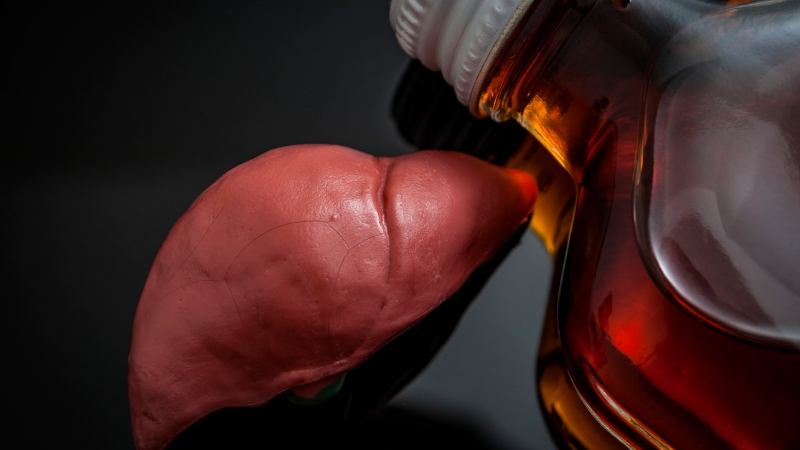
For decades, patients with liver disease related to alcohol use have been told they must be sober for six months before they can get a liver transplant. Many die before that six-month wait period is up. Now, a growing number of researchers are questioning that six-month waiting period.
In two published review papers, Johns Hopkins researchers outline the case for giving liver transplants to selected patients with alcoholic hepatitis. Their argument is backed by data from a six-year pilot study at Johns Hopkins, and they’ve now received an $8.4 million grant from the National Institutes of Health to expand the study to even more patients with alcoholic hepatitis.
“Alcoholic hepatitis patients have previously been stigmatized, and told that they don’t deserve this treatment that could save their life. Now, we’re trying to move forward and modernize our approach,” says Andrew Cameron, M.D., Ph.D., professor of surgery and chief of the division of transplantation at the Johns Hopkins University School of Medicine.Alcoholic hepatitis is inflammation of the liver caused by drinking excessive amounts of alcohol. As many as 50 percent of patients with severe forms of the disease die in just 28 days without treatment. While drugs such as steroids, as well as abstinence from drinking, can help resolve some cases of hepatitis, the only treatment for more advanced cases is liver transplant. However, due to concerns about patients returning to alcohol after their transplant, most liver transplant centers require six months of sobriety before allocating a liver to a person with alcoholic hepatitis. Relapsing alcoholism can cause problems with a new liver or a recurrence of hepatitis, and may also be associated with noncompliance in taking necessary post-transplant medications.
In 2012, however, a small study from France and Belgium published in the New England Journal of Medicine showed good outcomes in patients with alcoholic hepatitis who received liver transplants without a sobriety waiting period. In the wake of these results, Johns Hopkins launched a pilot study to waive the transplant waiting period for a selected group of patients with alcoholic hepatitis — those who are otherwise healthy, have a strong support network and good insight into the role that alcohol played in their disease.
Last year, the Johns Hopkins team reported on the outcomes of the first 46 patients who underwent liver transplantation under the pilot program, receiving livers between October 2012 and July 2017. Patients were followed for an average of 532 days after transplant. Psychiatric care was determined on an individual basis. During the follow-up period, the alcohol relapse rate among patients who did not have a wait period was identical to that seen in a group of 34 patients who received transplants under the standard six months of sobriety rule — in both cases, 28 percent relapsed at one point, but 98 percent of all patients were sober at the end of the study period. According to the National Institute on Alcohol Abuse and Alcoholism, around 90 percent of people in the general public with alcoholism relapse within four years of beginning a treatment. In addition, the rates of overall transplant functioning and patient survival were statistically identical between the groups and were what the researchers consider to be high, “better than that seen with other indications for liver transplant, such as hepatitis C,” adds Cameron.
Now, Cameron and his colleagues are calling for more transplant centers to follow Johns Hopkins’ lead. In review papers published this month and earlier this year in Journal of Intensive Care Medicine and Journal of Hepatology, they outline data seen in the patients at Johns Hopkins as well as a handful of other transplant centers with similar, smaller, pilot programs.
“I think what the field is looking for at this point is published results based on careful scientific research that help us answer who to transplant in a nonjudgmental and nonstigmatized way,” says Cameron. The Johns Hopkins group, he adds, with the largest existing cohort of patients with alcoholic hepatitis who have received transplanted livers, is in a position to provide this research. But more data is still needed on the long-term outcomes of these patients, what psychiatric interventions can help decrease relapse rates, as well as the underlying mechanisms of alcoholic hepatitis.
Many factors can increase a person’s risk for alcohol use disorder, says Mary E. McCaul, Ph.D., a Johns Hopkins professor of psychiatry and behavioral sciences who is collaborating with Cameron. “These factors generally are not under an individual’s control, and people do not choose to develop a drinking problem,” she says. “Importantly, people with an alcohol disorder can stop drinking and achieve a strong recovery; they should have equal access to lifesaving liver transplants.”
To answer some of the researchers’ outstanding questions, the National Institutes of Health awarded Cameron and his colleagues at Johns Hopkins an $8.4 million P50 grant to establish a new alcohol research center. Over the next five years, the researchers plan to continue with liver transplants in patients with alcoholic hepatitis when appropriate and compare these results with those transplanted after a six-month wait. Additionally, the center will help expand a liver tissue bank that researchers around the country can use to study alcoholic hepatitis, study why some people can drink more than others without getting liver disease, and answer ethical questions about transplanting livers in patients with alcoholic hepatitis.
“We used to view liver transplants in this population as a reward for what you’ve already done, for your sobriety in the past,” says Cameron. “Now, we’re looking at how we can allocate this limited resource based instead on what you can do in the future if you get a second chance.”
In addition to Andrew Cameron, other authors on the Journal of Hepatology paper are Gene Im of Icahn School of Medicine at Mount Sinai and Michael Lucey of University of Wisconsin. Other authors on the Journal of Intensive Care Medicine paper are Michelle Ma, Katie Falloon, Po-Hung Chen, Behnam Saberi, Aliaksei Pustavoitau, Elif Ozdogan, Zhiping Li, Benjamin Philosophe and Ahmet Gurakar, all from Johns Hopkins.
The authors received no financial support for the research or authorship of the review articles.
On the Web:
- Comprehensive Transplant Center
- Alcoholic Hepatitis | Health Library
