News Tips on Basic Research: Speeding Proteins and How Smell Affects Behavior
01/31/2019
ENZYME WARPS SPACE TO BREAK THE CELL’S SPEED LIMIT
Published Feb. 1 in Science
Johns Hopkins researchers have found that rhomboid enzymes, which are special proteins that cut other proteins, are able to break the “cellular speed limit” as they move through the cell membrane. Rhomboid enzymes do this by warping their surroundings, letting them glide quickly from one end of the membrane to another.
The cell membrane is a fatty layer that surrounds a cell and forms a border between the inside of the cell and the outside world. Nearly a third of our genes encode proteins that are needed at the border, making it a very crowded place through which it’s not easy to move—like a shopping mall before the holidays. Siniša Urban, Ph.D., professor of molecular biology and genetics at the Johns Hopkins University School of Medicine, labeled single rhomboid enzymes with chemicals that make them glow and discovered that they were moving much faster than they “should” be able to.In the 1970s, scientists found that the speed proteins move through the cell membrane follows a mathematical equation known as the Saffman-Delbrück viscosity limit. It takes into account the size and shape of the protein and the thickness, or viscosity, of the liquid the protein is moving through.
Reporting their findings in the journal Science, Urban’s team found that, instead of slowly trudging through the cell membrane, rhomboid enzymes zipped around quickly, moving twice as fast than predicted by the Saffman-Delbrück equation.
Looking more closely at the rhomboid enzymes, Urban’s team found that they interact with and change the shape of fats in the membrane as they move. The fats become less sticky, allowing the big protein to slip through.
Why do some proteins need to move quickly? The researchers believe that rhomboid enzymes developed this ability so they could scour the membrane quickly, looking for targets to cut. Some of these targets must be released swiftly from the membrane to provide real-time signals to other cells that conditions have changed. Other enzymes in the membrane probably also need to hurry their search for targets, and understanding how this happens in the rhomboid protein will help scientists learn more about these proteins and their roles in disease.
Rhomboid enzymes are found in the cell membrane of almost every life form on Earth and scientists are studying this enzyme superfamily as potential targets for treating parasitic infections, cancer, inflammation and neurodegeneration.
YOU KNOW THAT ‘RAIN’ SMELL? IT CAN HELP SCIENTISTS STUDY HOW SMELL AFFECTS BEHAVIOR
Published Aug. 7, 2018 in Cell Reports
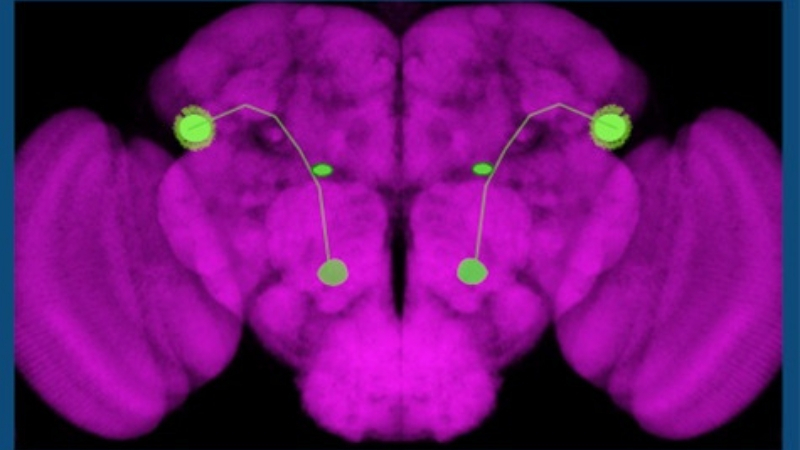
 |
The green shows neural circuits controlling the sense of smell in the brain of a common fruit fly, Drosophila melanogaster. |
Take a whiff when it rains after a dry spell, and breathe in the earthy scent that pervades the air. That smell comes from decomposing bacteria, and scientists call the compound geosmin.
Now, as reported in a paper published last summer in Cell, a team of researchers led by Chris Potter, Ph.D., an associate professor of neuroscience at the Johns Hopkins University School of Medicine, has used geosmin to create a new genetic technique in flies that could allow scientists to more accurately investigate how smell affects behavior.
They call the technique “olfactogenetics.” It works on the same principle as another popular neuroscience technique to genetically engineer specific neurons to fire off signals when hit by light. But instead of light, olfactogenetics uses geosmin.
Most odors are detected by multiple types of smell-sensing neurons, but geosmin is detected by only one type of neuron. That makes it easier for the researchers to program groups of neurons with the genetic code for sensing geosmin, while disabling it in those that already have the capability to detect geosmin. This way, when they release geosmin into the air, they can track which neurons are detecting the compound.
Because olfactogenetics uses an odor — not light — it allows the researchers to see how these neurons would normally function when sensing a smell, and it doesn’t run the risk of overstimulation.
