Case Study: Vascular Surgery
Case Study 1:
Revision Surgery for Thoracic and Abdominal Aortic Aneurysm Using a Minimally Invasive Endovascular Fenestrated Stent Graft
An 81-year old female patient (Jehovah’s Witness) presented with an expanding aortic aneurysm in the thoraco-abdominal (chest and abdomen) region that had enlarged from 5 to 7 centimeters.
The patient’s prior endovascular aortic aneurysm stent graft repair in the infra-renal portion of the abdominal aorta made the current aneurysm repair substantially more challenging. Since she is a Jehovah’s Witness and does not accept blood transfusions, it was decided that an open surgical approach was not an option due to the high risk of bleeding. A minimally invasive, endovascular repair was chosen as the ideal treatment method since the expected blood loss is decreased dramatically compared to traditional open aortic surgery.
The surgery was performed by one of our most experienced vascular surgeons, Dr. James Black, and took place over the course of 7 hours. The anesthesia care was provided by Dr. Steven Frank, the Director of the Center for Bloodless Medicine and Surgery at Johns Hopkins. The spinal drain performed nicely and the patient’s blood pressure was maintained above the threshold necessary to ensure blood flow to the kidneys and the spinal cord. Total blood loss was 300 ml (about 6% of total blood volume). The majority of the blood lost was returned to the patient using autologous blood salvage (Cell Saver). This option had been discussed with the patient, and she agreed to its use. She awoke from the surgery with normal movement in both arms and both legs.
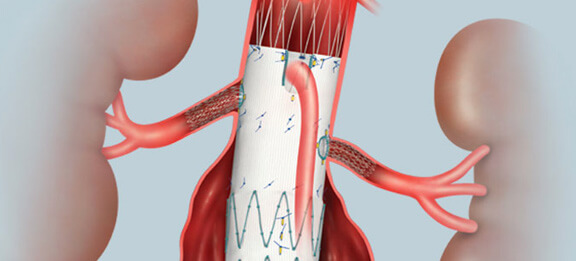 Fenestrated endovascular aortic stent graft
Fenestrated endovascular aortic stent graftIllustration permission for use by Cook Medical Incorporated, Bloomington, Indiana
The patient’s preoperative hemoglobin was 11.3 and the postoperative hemoglobin was 9.6, well within the margin of safety. Her postoperative course included 5 days of erythropoietin, intravenous iron, B12, and folate, a regimen recommended by our Bloodless team Hematology consultant, Dr. Linda Resar, in order to stimulate the bone marrow to produce red blood cells. After a few days of physical therapy, she was able to walk on her own and she was discharged from the hospital after an 8-day stay. Since discharge, she has not experienced any further problems, and has not needed to return.
Case Study 2:
Ascending Aortic Aneurysm Dissection Repair with Cardiopulmonary Bypass and Deep Hypothermic Circulatory Arrest
Blood conservation methods:
- EPO, IV iron, B12, Folic acid
- Intraoperative autologous normovolemic hemodilution (IANH)
- Retrograde autologous prime (RAP)
- Modified ultrafiltration
- Anti-fibrinolytic medication - Epsilon-aminocaproic acid (Amicar)
- Thromboelastography
- Minor fractions (cryoprecipitate and prothrombin complex concentrate (PCC)
A 37-year old male Jehovah’s Witness patient was transferred from an outside hospital to Johns Hopkins with a dissecting ascending aortic aneurysm. He presented with chest pain and a 15-cm aortic dissection diagnosed by CT scan. Given his history of prior cardiac surgery (aortic valve replacement), his aneurysm repair would require a “redo sternotomy” which is associated with substantially more blood loss than a first time surgery into the chest. He was treated with nitroprusside, a potent vasodilator, as well as other anti-hypertensive medications (Labetalol, Amlodipine) to keep his blood pressure down to avoid what might have been a catastrophic rupture of the aneurysm.
We elected to give him erythropoietin (EPO) 30,000 units/day, intravenous iron sucrose 200 mg/day, vitamin B12, and folic acid over a 1-week period, in order to increase his hemoglobin level prior to surgery from 12.7 to 13.7 g/dL. Given the challenging nature of the surgical procedure he required, we had to weigh the risk of aneurysm rupture vs. the risk of anemia, in choosing to wait on scheduling the surgery. Based on calculations of his red cell mass, our Bloodless Team Hematology consultant (Dr. Linda Resar) and his cardiac surgeon (Dr. Ashish Shah) made the decision to achieve a target hemoglobin close to 14 g/dL before attempting the surgery.
The intraoperative course went smoothly. A cardiac anesthesia team experienced with Jehovah’s Witness patients, led by Dr. Charles Brown, was assigned to provide the intraoperative care. A team of Perfusionists with Jehovah’s Witness experience (Larry Wolf and AnnMarie Fatula), were also assigned to the case. First 2 units of autologous blood were removed while maintaining a continuous connection to the patient. Intraoperative autologous normovolemic hemodilution (IANH) was then accomplished by giving volume expanders (albumin and crystalloids). Full cardiopulmonary bypass with retrograde autologous priming (RAP) was initiated using a “fem-fem” approach (femoral vein to femoral artery), which allowed us to avoid placing cannulas into the ascending aorta. The patient was cooled on cardiopulmonary bypass to 18 °C (about 10 °C lower temperature than the typical cardiac surgery case), in order to induce deep hypothermic circulatory arrest. This allowed the ascending aortic repair and a brief period (35 minutes) of total interruption of blood flow to the brain, which was required to complete the distal aortic anastomosis. The patient was then aggressively rewarmed to avoid bleeding from residual hypothermia during the postoperative period. During cardiopulmonary bypass, a hemoconcentrator (modified ultrafiltration) was used in order to provide concentrated whole blood for the patient and to minimize blood lost to the cardiopulmonary bypass circuit. Autologous blood salvage (the Cell Saver) was also used to return as much blood as possible to the patient from the bypass circuit. Epsilon-aminocaproic acid (Amicar) was used as an anti-fibrinolytic throughout the case to reduce bleeding.
Since the patient agreed to the minor blood fractions, coagulation was optimized using cryoprecipitate and 4-factor prothrombin complex concentrate (PCC). To reduce bleeding, coagulation was monitored by thromboelastography (TEG) both during and after surgery. Postoperatively, the patient awoke without any neurologic deficits. He was discharged from the ICU within 24 hours following surgery with a hemoglobin of 11.1 g/dL and a platelet count of 210K. Postoperative bleeding was minimal. The patient’s wife and family members were incredibly grateful for the care he received.
