News Tips on Basic Research: What Makes Cells Move?
03/21/2019
Published Sept. 07 in Proceedings of the National Academies of Sciences
Your cells are on the move. Immune cells rove your body hunting for pathogens; cells organize to assemble complex organs; skin cells pull together to heal wounds; and rogue cancer cells migrate and metastasize. Without this remarkable ability to coordinate movement, cells could not divide or reach their proper place in the body. This leads to diseases, such as cardiovascular disorders, Parkinson's disease and cancer.
But how do cells get from one place to another? To understand this, Johns Hopkins researchers looked to a single-celled amoebae called Dictyostelium discoideum commonly found in soil. Their results, say the researchers, could shed more light on biological processes that depend on moving cells.
Most cells inch themselves along through a series of signals that instruct the front end to pull forward with footlike extensions, called pseudopods, and the back end to contract and push.
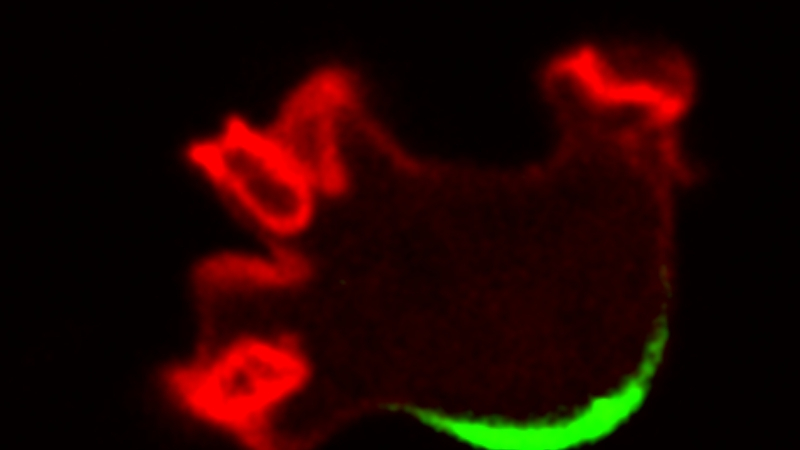 |
Dictyostelium amoebae moving under a microscope. The front of the cell is labeled in red; the back, in green. |
Cells balance these pushing and pulling forces so that their movements do not spiral out of control. The balance, the researchers say, happens at a series of chemical “checkpoints,” where the cell receives feedback to adjust its motion. “Without this ability to self-regulate themselves through this feedback, the cells would make protrusions that would not be efficient for movement and would never reach their destination,” says Xiaoguang Li, first author on the study and a graduate student in the laboratory of Peter Devreotes, Ph.D., professor of cell biology at The Johns Hopkins University School of Medicine.
Li turned her attention to one of these checkpoints, a fatty molecule called phosphatidylinositol 3,4-bisphosphate (PI(3,4)P2), which is an important component of cell membrane. When cells move, PI(3,4)P2 slides to the back of the cell and helps push the cell forward. So, what does PI(3,4)P2 do? A common way to find out is to shut off the molecule to see how the cell behaves. Li genetically engineered Dictyostelium cells to block production of PI(3,4)P2. Without the molecule, the cells became hyperactive and disorganized with helter-skelter protrusions and exaggerated spreading.
“It looked like we flipped the switch between controlled movement and a frenzied swarm,” says Li.
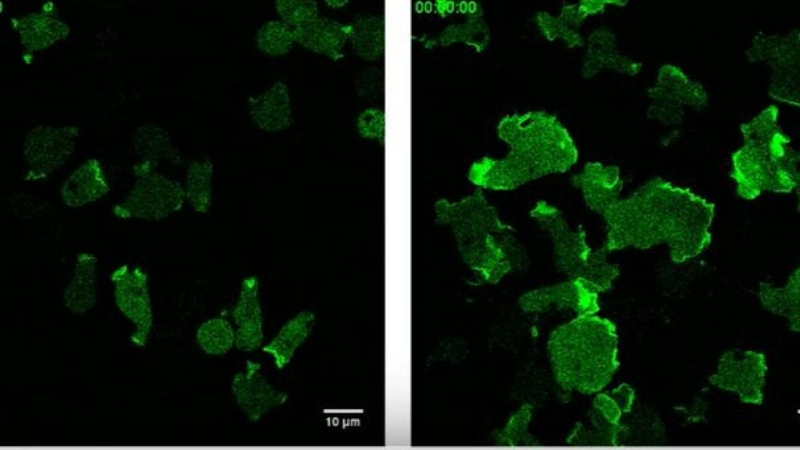
When the checkpoint is disabled by a genetic mutation, the cells become hyperactive and move erratically. Left video, checkpoint intact; Right video, mutated checkpoint. |
In the hyperactive cells that lacked PI(3,4)P2, Li found a substantial increase in the activity of another protein, called Ras, which is one of the most important players in cancer. While Ras is activated at the front of moving cells and helps the cells create the footlike extensions that help pull it forward, PI(3,4)P2 is distributed to the back in a complementary manner.
In additional experiments, Li, along with postdoctoral fellow Marc Edwards, have investigated the molecular mechanisms that bring about the inhibitory interactions between Ras activation and PI(3,4)P2. In the future, Li would like to investigate what controls PI(3,4)P2 pools in the cells. The long-term goal is to have a complete description of the network controlling migratory behavior, which will provide new targets for drugs that could be applied for cell migration.
