Research Study on Bleeding in Scoliosis Surgeries Lays Groundwork for Future Discovery
Researchers at Johns Hopkins All Children’s Hospital published a study that investigated the factors that contribute to blood loss during scoliosis surgery.
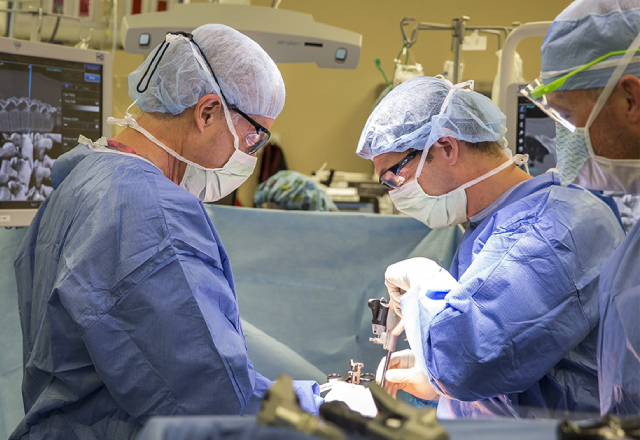
For children with idiopathic scoliosis, an abnormal curvature of the spine, surgery is necessary if the curve is severe enough. The surgery can involve quite a bit of bleeding and it’s often not clear why one child may bleed more than another.
Researchers at Johns Hopkins All Children’s Hospital published a study that investigated the factors that contribute to blood loss during scoliosis surgery. Published in the journal Pediatric Anesthesia, the study lays the groundwork for future efforts to develop a predictive model to help physicians identify patients at a higher risk for bleeding and develop new ways to reduce their risk.
“It’s a patient population that we see very frequently at Johns Hopkins All Children’s and many other pediatric hospitals,” says Allison Fernandez, M.D., pediatric anesthesiologist and lead author on the study.
Scoliosis is a condition in which the spine is curved – on an X-ray it looks more like an “S” or a “C” than a straight line. Idiopathic scoliosis is one of several types; “idiopathic” means the cause is unknown. It’s the most common form of scoliosis and is more common in girls.
Treatment for scoliosis often first involves observation and the use of a brace to keep the spinal curve from progressing. However, surgery is recommended when the curve of the spine progresses beyond 40 to 45 degrees. Left untreated, it can cause chronic pain, deformity, reduced lung function and other health problems.
Spinal fusion surgery for scoliosis involves correcting the spinal deformity by placing two metal rods onto the spine. The rods are attached to screws that have been placed into each level of the spine and act as anchors for the rods. The rods hold the spine straight in the frontal plane but are bent to create the appropriate alignment as viewed from the side, so that the spine will fuse in the correct alignment.
The surgical and anesthesia teams take steps to minimize bleeding, such as safely lowering the patient’s blood pressure during surgery, and using electrocautery, which serves to seal off small blood vessels as tissues are being cut as part of a surgical procedure. Nevertheless, scoliosis surgery can involve a lot of bleeding – though it doesn’t always. Before a child receives surgery for scoliosis, certain laboratory blood tests are run, looking at certain common factors that influence bleeding and clotting, to determine if that particular patient is at a higher risk for bleeding during the surgery.
While these lab tests may help to exclude patients who are born with relatively rare bleeding disorders that may not be obvious in the absence of a prior history of surgery or other significant “challenge” to the clotting system, they don’t accurately predict bleeding risk outside of those diseases.
The patients (who, along with their parents, agreed for them to participate in the study) were all adolescents with scoliosis, who did not have other conditions like cerebral palsy or muscular dystrophy. A sample of blood was obtained from each patient pre-operatively and stored in the hospital’s biorepository, which maintains stored blood and tissue samples from patients who have agreed to participate in research studies that involve collections of these samples. The team ran tests on the blood to look for factors that both tied to instances where the patient experienced more bleeding during surgery, and others where the patient did not.
The study is the first original research publication to come from the hospital’s iPICS (Institution-wide Prospective Inception Cohort Study) initiative, which launched in 2013. iPICS aims to identify factors that affect the progress and outcomes in children with a variety of acute and chronic health conditions, with the goal of developing more tailored treatment approaches.
“If we can achieve better risk stratification within a given condition, then we can develop more precise therapies,” says Neil Goldenberg, M.D., Ph.D., professor of pediatrics and medicine, director of the Johns Hopkins All Children’s Institute for Clinical and Translational Research, and associate dean for Research at Johns Hopkins All Children’s. “When we identify, based on data, which children are at highest risk for a certain adverse outcome (such as relapse or worsening of a disease), we can then study a new intervention — a new drug, for example, or a new way of using existing therapies — with the goal of preventing relapse or worsening of disease; in this way, we can improve disease outcomes for the future.”
The research team looked at a number of factors that could affect bleeding, such as surgical factors like length of surgery, and demographic features like age and gender.
In regard to the blood clotting system, Goldenberg explains, “Apart from the integrity of tissues and vessels (which is difficult to measure), how much bleeding you have is a reflection not only of how well your blood clots but also how well those clots stay intact.”
The routine pre-op labs don’t measure this function of clot stability and breakdown, called “fibrinolysis;” however, a research assay (test) that Goldenberg developed with colleagues in 2003 measures both the clot formation and fibrinolysis abilities of blood, called the Clot Formation and Lysis (CloFAL) assay. The assay has been applied in several previous published studies in children and adults with a variety of diseases; iPICS was the first study in which it has been applied in children undergoing surgery for scoliosis.
The research team put all of the data – including surgical factors, demographic data, routine pre-op lab results and results of the CloFAL assay – into a model. Their results showed that none of the routine or historically studied factors were predictive of bleeding severity, but the CloFAL assay was.
As this was a single-center study limited to patients from Johns Hopkins All Children’s, the next step would be to validate the results at other hospitals and further investigate other factors that influence bleeding, eventually leading to an interventional study to determine if a new approach, such as a new drug or therapy, would be effective in lowering the bleeding risk in high-risk pediatric patients.
The team’s work was published in the journal Pediatric Anesthesia. The team involved in the publication included Fernandez; Neustadt; Hahn; Anthony Nguyen, biostatistician; Ernest Amankwah, Ph.D., epidemiologist and assistant professor in oncology; and Goldenberg.
With involvement from across the hospital – including anesthesia, orthopaedic surgery, hematology, biostatistics/epidemiology, the biorepository team, study coordinators from the research operations unit, and laboratory technologists in the research laboratory facility of the Johns Hopkins All Children’s Research and Education Building – the study reflects the multidisciplinary nature of science and the power of a campus that brings together multiple experts in a hub of research and innovation.
Currently, researchers at Johns Hopkins All Children’s are designing additional studies similar to this one in other scoliosis patients, that hope to also test the clotting system in greater depth, toward identifying additional factors that may serve as targets for future treatments.
Gregory Hahn, M.D., is on the medical staff of Johns Hopkins All Children’s Hospital, Inc. (“JHACH”), but is an independent practitioner, not an employee or agent of JHACH.
