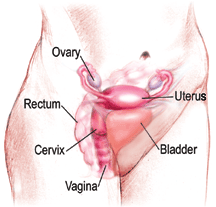
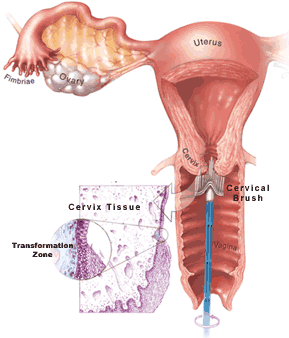
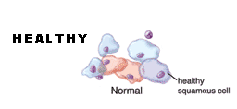
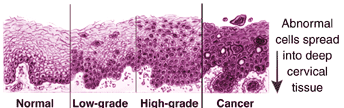
A normal Pap smear shows healthy squamous cells (flat cells that look like fish scales) from the surface of the cervix. There are no signs of infection and no abnormal cells. Even if your Pap results are healthy, you should be tested regularly. The tests screen for infectious agents that may be harmful if allowed to persist. Early detection of any infection will allow for better treatment and will help you maintain your health.
Abnormal Pap test results include:
ASCUS ~ Atypical squamous cells of undetermined significance
This diagnosis means that some of the cells on your Pap smear did not look entirely normal but did not meet diagnostic criteria for a lesion. Your doctor may either repeat your Pap smear, or perform a colposcopy. The lab may test your Pap smear specimen for HPV.
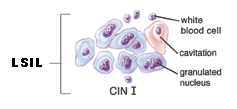
LSIL ~ Low-grade squamous intraepithelial lesion
This diagnosis means there are early changes in the size and shape of the cells. LSILs are often associated with HPV, which may also cause genital warts. These lesions, in women with intact immune systems, often resolve without intervention within 18 to 24 months. Low-grade lesions may also be called mild dysplasia, or CIN1. If it is your first abnormal Pap smear, your doctor will likely recommend a colposcopy.
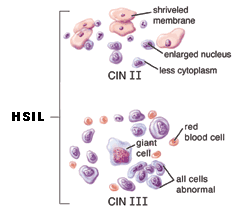
HSIL ~ High-grade squamous intraepithelial lesion
This diagnosis means the cells appear very different from normal cells. These precancerous lesions are more severe than with LSIL, but involve cells on the surface of the cervix. They may also be called moderate or severe dysplasia, or CIN 2 or 3. The treatment for HSIL is to remove the abnormal tissue. This can be done in several ways. See the treatment section for more information.