Specialty Areas
Find the treatment you need Otolaryngology-Head and Neck Surgery Specialty Areas
-
Audiology/ Hearing Disorders
Our team provides: hearing testing, hearing aid selection and fitting and implantable hearing devices for patients of all ages.

-
Cochlear Implant and Rehabilitation
Using cochlear implants, our team provides the medical, technical, and rehabilitative resources to enhance our patients' connection with spoken language.

-
Oral & Maxillofacial Surgery and Dentistry
Offering oral surgery services along with dentistry and cosmetic and hygiene procedures, including wisdom tooth extraction, dental implants, jaw surgery and veneers.

-
Eustachian Tube Health Center
The Johns Hopkins Eustachian Tube Health Center provides comprehensive evaluation, diagnosis and management of Eustachian tube dysfunction that include medical, multidisciplinary and surgical approaches.
-
Facial Nerve Schwannoma Center
The Facial Nerve Schwannoma Center offers a comprehensive multi-specialty approach and expertise in the management and treatment of facial nerve schwannomas.

-
Facial Plastic and Reconstructive Surgery
Performing both cosmetic and reconstructive procedures, our surgeons consistently seek to advance the field of facial plastic surgery.
-
General Ear Nose and Throat (ENT) Care
We provide introductory ENT evaluations and care for adult and pediatric patients even if your symptoms are vague or unclear symptoms.

-
Head and Neck Tumor Center
Treating cancers that affect areas such as nasal passages, sinuses and the throat, our team has expertise in head and neck surgery, radiation oncology, medical oncology and endocrinology.

-
Laryngeal and Airway Disorders
Expert care for patients who have airway, voice, swallowing and laryngeal problems that require specialized assistance.

-
Otology and Neurotology
This multidisciplinary team of experts is dedicated to treating acoustic neuromas and other tumors of the skull base.

-
Pediatric Otolaryngology (ENT)
Specialized care for children and adolescents with such illnesses as tonsillitis, chronic and recurring ear infections, head and neck masses, and sleep apnea.

-
Salivary Gland Center
Providing state-of-the-art evaluation, diagnosis, and surgical treatment for patients with salivary gland tumors and other salivary gland disorders.
-
Sinus Center
Focusing on diagnosis and treatment of conditions of the nose and paranasal sinuses including allergies, septal deviations, chronic rhinosinusitus, neoplasms and Graves' Orbitopathy.

-
Skull Base Tumors
Specializing in the treatment of skull-base tumors, which grow along various regions of the bottom part of the skull, using innovative and minimally invasive techniques.
-
Snoring and Sleep Surgery
Comprehensive care for patients with obstructive sleep apnea or snoring who have not had successful medical treatment or are interested in exploring other treatment options.

-
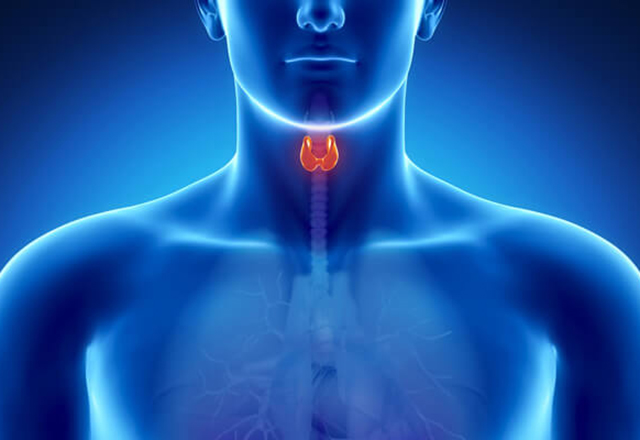
Thyroid and Parathyroid Conditions
Focused on diagnosing and treating patients with a variety of thyroid and parathyroid conditions including tumors, nodules and hyperparythroidism.