Research Story Tip: Johns Hopkins Team Shows Nervous and Immune Systems ‘Need to Talk’ For Bone Repair
06/18/2020
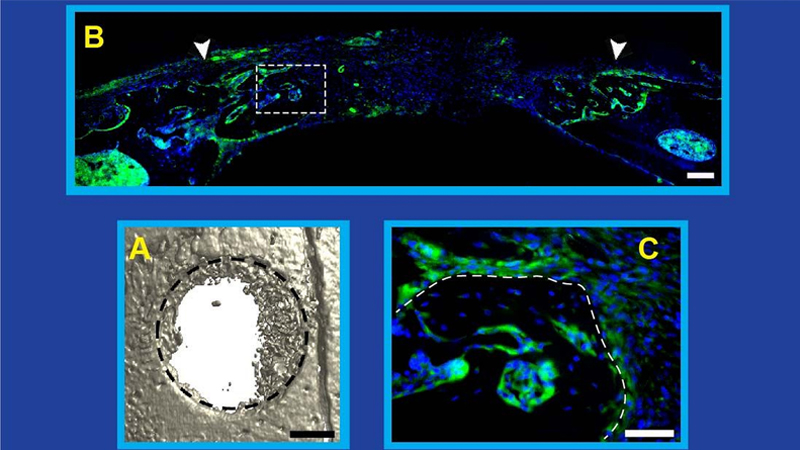
In a December 2019 study, a team of Johns Hopkins Medicine researchers demonstrated in mice that repair of bone fractures requires the generation, growth and spread of nerve cells, or neurons, throughout the injured area. This, they showed, partly relies on a protein known as nerve growth factor (NGF). Now, the researchers have dug deeper into this process to better understand how the nervous and immune systems work together with NGF to enable nerve regrowth during bone repair.
In a new study, published in the May 26, 2020, issue of the journal Cell Reports, the researchers found once again in mice that two proteins — tropomyosin receptor kinase-A (TrkA) and NGF — bind together to stimulate innervation (the supplying of nerves), and subsequently, new bone at an injured site. What surprised them was that the NGF that mattered most in this process came from an unexpected source: macrophages, the white blood cells that alert the immune system to foreign invaders through inflammation, and then engulf and remove the attackers from the body.
“Previous research has shown that immune cells are clearly important in bone repair, but what we determined in our study is that macrophages and their inflammatory signals also kickstart nerve regrowth in injured bone,” says Aaron James, M.D., Ph.D., associate professor of pathology at the Johns Hopkins University School of Medicine and co-senior author of both studies.
In other words, James explains, the team’s experiments revealed “that NGF-TrkA signaling is how macrophages ‘talk’ to nerve fibers so that bone healing can begin.”
When bones are injured, there is a large release of the NGF neurotrophin (a protein that induces the survival, development and function of neurons). This activates sensory nerves to grow into the injured tissue. These sensory nerves play multiple roles, including alerting the body through pain that the bone is broken and regulating the healing process.
To define the mechanism by which bone is repaired, the researchers removed the same small piece of skull from each of the mice in the study. By manipulating various steps of the NGF-TrkA signaling pathway in different mice, the team found that: (1) the release of NGF coincides with the beginning of innervation, (2) bone injury stimulates the increased production of NGF, (3) inflammation at the injury site drives NGF production by macrophages (which are drawn by chemical signals released during inflammation), (4) increased amounts of NGF elicit new nerve formation in the injured tissue, (5) disrupting the production of NGF reduces innervation and impairs calvarial bone regeneration, and (6) NGF produced by macrophages is the neurotrophin required for bone repair.
“We now understand that nerve growth and bone repair are linked processes,” James says. “Knowing this, we may be able to find ways to maximize our innate healing capacities. Developing new methods to improve bone healing would greatly benefit many people, especially the elderly, where injuries such as hip fractures often lead to worse outcomes than heart attacks.”
