Age-Related Macular Degeneration (AMD)
What You Need to Know
- Age-related macular degeneration (AMD) is a disease that affects a person’s central vision.
- AMD can result in severe loss of central vision, but people rarely go blind from it.
- Risk factors for AMD include being 50 and older, smoking, having high blood pressure and eating a diet high in saturated fat.
- Although there is no cure for AMD, there are treatment options that may prevent or slow the progression of the disease.
Age-Related Macular Degeneration Definition
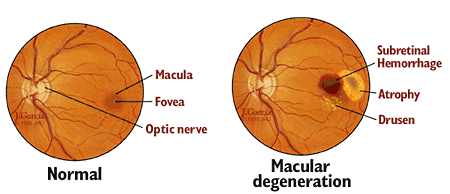
Age-related macular degeneration is the most common cause of severe loss of eyesight among people 50 and older. Only the center of vision is affected with this disease. It is important to realize that people rarely go blind from it.
AMD affects the central vision, and with it, the ability to see fine details. In AMD, a part of the retina called the macula is damaged. In advanced stages, people lose their ability to drive, to see faces, and to read smaller print. In its early stages, AMD may have no signs or symptoms, so people may not suspect they have it.
Types of Age-Related Macular Degeneration and Causes
The two primary types of age-related macular degeneration have different causes:
-
Dry. This type is the most common. About 80% of those with AMD have the dry form. Its exact cause is unknown, although both genetic and environmental factors are thought to play a role. This happens as the light-sensitive cells in the macula slowly break down, generally one eye at a time. The loss of vision in this condition is usually slow and gradual. It is believed that the age-related damage of an important support membrane under the retina contributes to dry age-related macular degeneration.
-
Wet. Though this type is less common, it usually leads to more severe vision loss in patients than dry AMD. It is the most common cause of severe loss of vision. Wet AMD happens when abnormal blood vessels start to grow beneath the retina. They leak fluid and blood — hence the name wet AMD — and can create a large blind spot in the center of the visual field.
Risk Factors for Age-Related Macular Degeneration
There are several risk factors that can contribute to developing age-related macular degeneration, including:
-
Being 50 and older
-
Eating a diet high in saturated fat
-
Smoking
Age-Related Macular Degeneration Symptoms
The following are the most common symptoms of age-related macular degeneration. However, each individual may experience symptoms differently. Symptoms may include:
-
Blurry or fuzzy vision
-
Difficulty recognizing familiar faces
-
Straight lines appear wavy
-
A dark, empty area or blind spot appears in the center of vision
-
Loss of central vision, which is necessary for driving, reading, recognizing faces and performing close-up work
The presence of drusen, which are tiny yellow deposits in the retina, is one of the most common early signs of age-related macular degeneration. It may mean the eye is at risk for developing more severe age-related macular degeneration. These will be visible to your doctor during an eye exam.
The symptoms of age-related macular degeneration may look like other eye conditions. Speak with an eye care professional for diagnosis.
Research Shows AI Used to Predict Disease Progression

Age-Related Macular Degeneration Diagnosis
In addition to a complete medical history and eye exam, your eye doctor may do the following tests to diagnose age-related macular degeneration:
-
Visual acuity test. This common eye chart test measures vision ability at various distances.
-
Pupil dilation. The pupil is widened with eyedrops to allow a close-up examination of the eye’s retina.
-
Fluorescein angiography. Used to detect wet age-related macular degeneration, this diagnostic test involves a special dye injected into a vein in the arm. Pictures are then taken as the dye passes through the blood vessels in the retina, helping the doctor evaluate if the blood vessels are leaking and whether or not the leaking can be treated.
-
Amsler grid. Used to detect wet age-related macular degeneration, this test uses a checkerboardlike grid to determine if the straight lines in the pattern appear wavy or missing to the patient. Both indications may signal the possibility of age-related macular degeneration.
Amsler Grid
To use the Amsler grid, follow these steps:
- Wearing any glasses you normally use to read, hold the grid 12 to 15 inches away from your face in good light.
- Cover one eye.
- Look directly at the center dot with your uncovered eye and keep your eye focused on it.
- While looking directly at the center dot, notice in your side vision if all grid lines look straight or if any lines or areas look blurry, wavy, dark or blank.
- Follow the same steps with the other eye.
If you notice any areas of the grid that appear darker, wavy, blank or blurry, contact your ophthalmologist right away.

Age-Related Macular Degeneration Treatment
Specific treatment for age-related macular degeneration will be determined by your doctor based on:
-
Your age, overall health and medical history
-
Extent and nature of the disease
-
Your tolerance for specific medications, procedures or low-vision therapies
-
Expectations for the course of the disease
-
Your opinion or preference
Currently, there is no treatment for dry age-related macular degeneration, though vision rehabilitation programs and low-vision devices can be used to build visual skills, develop new ways to perform daily living activities and adjust to living with age-related macular degeneration.
The main treatment for wet AMD is the injection of medications called anti-VEGF agents. VEGF stands for vascular endothelial growth factor. A high level of VEGF in the eye is linked to the formation of the abnormal blood vessels that cause much of the damage in wet AMD. Anti-VEGF agents are used to combat the disease process and reduce the damaging effects of these leaky abnormal blood vessels. They are also able to effectively stabilize vision in many patients.
In some patients, anti-VEGF injections actually improve the level of visual acuity. Anti-VEGF medications are administered by injecting them directly into the affected eye. Although this sounds daunting, the procedure is done with a very fine needle and under the cover of numbing (anesthetic) eyedrops, so patients are usually very comfortable. Anti-VEGF treatment is usually administered regularly over time, requiring multiple injections to maintain the treatment effect, and your retinal physician will discuss the best treatment schedule for you. In selected patients, other treatments, such as laser therapy, can be used, if necessary.
Complications of Age-Related Macular Degeneration
Age-related macular degeneration can result in severe loss of central vision but rarely causes blindness. It can, however, make it difficult to read, drive or perform other daily activities that require fine central vision. In AMD, the health of the peripheral retina is unaffected, so patients can rest assured that their peripheral (side) vision, and their ability to walk around without bumping into things, is usually preserved.
Treatment Get the Care You Need
The macular degeneration experts of Wilmer Eye Institute’s retina division provide care at seven convenient locations throughout Maryland.





