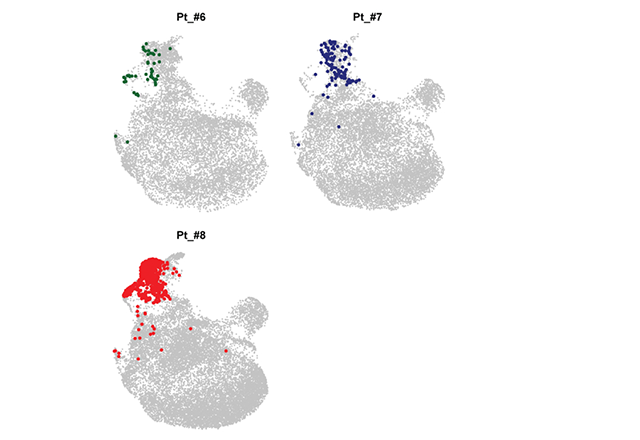
Latest Cancer News
-
Our Experts
The Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins includes a wide array of medical professionals and laboratory scientists. Many of our experts are nationally and internationally recognized as leaders in the research and treatment of cancer.

-
Cancers We Treat
At the Johns Hopkins Kimmel Cancer Center, our experts are dedicated to providing our patients the best treatment and quality of care possible. Patients are seen by multi-disciplinary teams for individualized treatment plans.
-
Locations
At the Johns Hopkins Kimmel Cancer Center, our experts are dedicated to providing our patients the best treatment and quality of care possible. Patients are seen by multi-disciplinary teams for individualized treatment plans.
-
Johns Hopkins and the National Cancer Plan
Imagine a world where most cancers can be prevented. What does that mean for you? The National Cancer Plan, led by the National Cancer Institute, is a call to action for each of us to do our part to help prevent more cancers and ensure that every person with cancer lives a full and active life. Explore the plan’s eight goals to end cancer.

-
Kimmel in the Community
The Johns Hopkins Kimmel Cancer Center is in your community. Community outreach and education combines our center's programs with our community partners to bring the best information in cancer treatment. Learn more about our community outreach and education activities.

-
Patient and Family Services
The Harry J. Duffey Family Patient and Family Services Program offers a variety of resources to assist during this time, such as emotional support, assistance with temporary housing and transportation and aid in managing care.

Featured Publications
Request An Appointment
Before Making the Call
410-955 8964
The Johns Hopkins Hospital
1-855-695-4872
Outside of Maryland (toll free)
+1-443-219-6052
International